C-1027
C-1027 or Lidamycin is an antitumor antibiotic consisting of a complex of an enediyne chromophore and an apoprotein.[2][3][4][5] [6][7] It shows antibiotic activity against most Gram-positive bacteria.[8] It is one of the most potent cytotoxic molecules known, due to its induction of a higher ratio of DNA double-strand breaks than single-strand breaks.
 | |
| Names | |
|---|---|
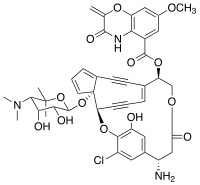
| IUPAC name
(3R,4R,14R,19S)-22-chloro-4-{[(2S,3R,4R,5S)-5-(dimethylamino)-3,4-dihydroxy-6,6-dimethyloxan-2-yl]oxy}-23-hydroxy-14-(3-hydroxy-7-methoxy-2-methylidene-2H-1,4-benzoxazine-5-carbonyloxy)-17-oxo-2,16-dioxapentacyclo[18.2.2.1⁹,¹³.0³,¹⁰.0⁴,⁸]pentacosa-1(22),5,7,9,11,13(25),20,23-octaen-19-aminium | |
| Other names
Lidamycin chromophore | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
PubChem CID |
|
| |
| |
| Properties | |
| C43H42ClN3O13 | |
| Molar mass | 844.267 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
C-1027's chromophore contains a nine-membered enediyne that is responsible for most of the molecule's biological activity.[8] Unlike other enediynes, this molecule contains no triggering mechanism. It is already primed to undergo the cycloaromatization reaction without external activation to produce the toxic 1,4-benzenoid diradical species. C-1027 can induce oxygen-independent interstrand DNA crosslinks in addition to the oxygen-dependent single- and double-stranded DNA breaks typically generated by other enediynes. This unique oxygen-independent mechanism suggests that C-1027 may be effective against hypoxic tumor cells.[9]
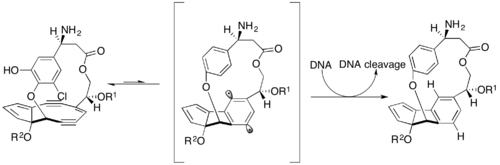
C-1027 shows promise as an anticancer drug and is currently undergoing phase II clinical trials in China,[10] with a 30% success rate.[11] It can induce apoptosis in many cancer cells and recent studies have indicated that it induces unusual DNA damage responses to double-strand breaks, including altering cell cycle progression and inducing chromosomal aberrations.[7]
References
- Pubchem. "Lidamycin". pubchem.ncbi.nlm.nih.gov. Retrieved 5 May 2018.
- Hu, Jilan; Xue, Yu-Chuan; Xie, Mei-Yu; Zhang, Rui; Otani, Toshio; Minami, Yoshinori; Yamada, Yuji; Marunaka, Teruyoshi (1988). "A new macromolecular antitumor antibiotic, C-1027. I. Discovery, taxonomy of producing organism, fermentation and biological activity". The Journal of Antibiotics. 41 (11): 1575–1579. doi:10.7164/antibiotics.41.1575. PMID 3198491.
- Otani, Toshio; Minami, Yoshinori; Marunaka, Teruyoshi; ZHANG, Rui; Xie, Mei-Yu (1988). "A new macromolecular antitumor antibiotic, C-1027. II. Isolation and physico-chemical properties". The Journal of Antibiotics. 41 (11): 1580–1585. doi:10.7164/antibiotics.41.1580. PMID 3198492.
- Zhen, Yong-Su; Ming, Xiu-Ying; Yu, Bin; Otani, Toshio; Saito, Hitoshi; Yamada, Yuji (1989). "A new macromolecular antitumor antibiotic, C-1027. III. Antitumor activity". The Journal of Antibiotics. 42 (8): 1294–1298. doi:10.7164/antibiotics.42.1294. PMID 2759910.
- Ken-ichiro, Yoshida; Minami, Yoshinori; Azuma, Ryotaro; Saeki, Mayuko; Otani, Toshio (1993). "Structure and cycloaromatization of a novel enediyne, C-1027 chromophore". Tetrahedron Lett. 34 (16): 2637–2640. doi:10.1016/S0040-4039(00)77644-1.
- Liang ZX (April 2010). "Complexity and simplicity in the biosynthesis of enediyne natural products". Natural Product Reports. 27 (4): 499–528. doi:10.1039/b908165h. PMID 20336235.
- Zhen YZ, Lin YJ, Li Y, Zhen YS (July 2009). "Lidamycin shows highly potent cytotoxic to myeloma cells and inhibits tumor growth in mice". Acta Pharmacologica Sinica. 30 (7): 1025–32. doi:10.1038/aps.2009.75. PMC 4006655. PMID 19575006.
- Xu YJ, Zhen YS, Goldberg IH (May 1994). "C1027 chromophore, a potent new enediyne antitumor antibiotic, induces sequence-specific double-strand DNA cleavage". Biochemistry. 33 (19): 5947–54. doi:10.1021/bi00185a036. PMID 8180224.
- Chen Y, Yin M, Horsman GP, Shen B (March 2011). "Improvement of the enediyne antitumor antibiotic C-1027 production by manipulating its biosynthetic pathway regulation in Streptomyces globisporus". Journal of Natural Products. 74 (3): 420–4. doi:10.1021/np100825y. PMC 3064734. PMID 21250756.
- Wang L, Wang S, He Q, Yu T, Li Q, Hong B (August 2012). "Draft genome sequence of Streptomyces globisporus C-1027, which produces an antitumor antibiotic consisting of a nine-membered enediyne with a chromoprotein". Journal of Bacteriology. 194 (15): 4144. doi:10.1128/JB.00797-12. PMC 3416545. PMID 22815456.
- Shen B, Yan X, Huang T, Ge H, Yang D, Teng Q, Rudolf JD, Lohman JR (January 2015). "Enediynes: Exploration of microbial genomics to discover new anticancer drug leads". Bioorganic & Medicinal Chemistry Letters. 25 (1): 9–15. doi:10.1016/j.bmcl.2014.11.019. PMC 4480864. PMID 25434000.