Teriflunomide
Teriflunomide, sold under the brand name Aubagio, is the active metabolite of leflunomide.[2] Teriflunomide was investigated in the Phase III clinical trial TEMSO as a medication for multiple sclerosis (MS). The study was completed in July 2010.[3] 2-year results were positive.[4] However, the subsequent TENERE head-to-head comparison trial reported that "although permanent discontinuations [of therapy] were substantially less common among MS patients who received teriflunomide compared with interferon beta-1a, relapses were more common with teriflunomide."[5] The drug was approved for use in the United States in September 2012[6][7] and for use in the European Union in August 2013.[8]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Aubagio |
| Other names | A77 1726 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a613010 |
| License data |
|
| Pregnancy category | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | >99.3% |
| Elimination half-life | 2 weeks |
| Excretion | Biliary/fecal, renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.170.077 |
| Chemical and physical data | |
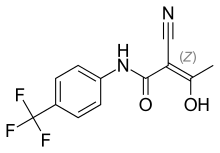
| Formula | C12H9F3N2O2 |
| Molar mass | 270.211 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mechanisms of action
Teriflunomide is an immunomodulatory drug inhibiting pyrimidine de novo synthesis by blocking the enzyme dihydroorotate dehydrogenase. It is uncertain whether this explains its effect on MS lesions.[9]
Teriflunomide inhibits rapidly dividing cells, including activated T cells, which are thought to drive the disease process in MS. Teriflunomide may decrease the risk of infections compared to chemotherapy-like drugs because of its more-limited effects on the immune system.[10]
It has been found that teriflunomide blocks the transcription factor NF-κB. It also inhibits tyrosine kinase enzymes, but only in high doses not clinically used.[11]
Activation of leflunomide to teriflunomide
The branded drug teriflunomide is the main active in vivo metabolite of the generically available leflunomide. Upon administration of leflunomide, 70% of the drug administered converts into teriflunomide. The only difference between the molecules is the opening of the isoxazole ring. This is considered a simple structural modification and a technically simple one-step synthetic transformation. Upon oral administration of leflunomide in vivo, the isoxazole ring of leflunomide is opened and teriflunomide is formed.[12]
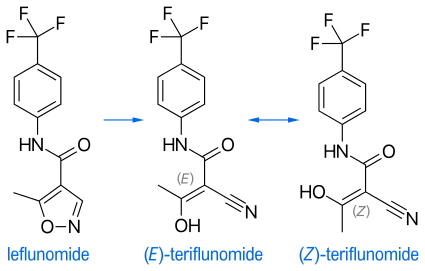
"Regardless of the substance administered (leflunomide or teriflunomide), it is the same molecule (teriflunomide)—the one exerting the pharmacological, immunological or metabolic action in view of restoring, correcting or modifying physiological functions, and does not present, in clinical use, a new chemical entity to patients."[12] Because of this, EMA initially had not considered teriflunomide being a new active substance.[15]
See also
See leflunomide for information on pharmacokinetics, side effects, contraindications and other data.
References
- "Teriflunomide (Aubagio) Use During Pregnancy". Drugs.com. 11 September 2019. Retrieved 2 March 2020.
- Magne D, Mézin F, Palmer G, Guerne PA (November 2006). "The active metabolite of leflunomide, A77 1726, increases proliferation of human synovial fibroblasts in presence of IL-1beta and TNF-alpha". Inflammation Research. 55 (11): 469–75. doi:10.1007/s00011-006-5196-x. PMID 17122964.
- ClinicalTrials.gov Phase III Study of Teriflunomide in Reducing the Frequency of Relapses and Accumulation of Disability in Patients With Multiple Sclerosis (TEMSO)
- "Sanofi-Aventis' Teriflunomide Comes Up Trumps in Two-Year Phase III MS Trial". 15 Oct 2010.
- Gever J (June 4, 2012). "Teriflunomide Modest Help but Safe for MS". medpage. Joint meeting of the Consortium of Multiple Sclerosis Centers and the Americas Committee for Treatment and Research in Multiple Sclerosis. Retrieved June 4, 2012.
- "Drug Approval Package: Aubagio (teriflunomide) Tablets NDA #202992". U.S. Food and Drug Administration (FDA). 5 November 2012. Retrieved 1 March 2020.
- "FDA approves new multiple sclerosis treatment Aubagio" (Press release). U.S. Food and Drug Administration (FDA). Archived from the original on 2012-09-13. Retrieved 2012-09-14.
- "Aubagio EPAR". European Medicines Agency (EMA). 26 February 2020. Retrieved 1 March 2020.
- Spreitzer H (March 13, 2006). "Neue Wirkstoffe - Teriflunomid". Österreichische Apothekerzeitung (in German) (6/2006).
- Vollmer T (May 28, 2009). "MS Therapies in the Pipeline: Teriflunomide". EMS News (May 28, 2009).
- Breedveld FC, Dayer JM (November 2000). "Leflunomide: mode of action in the treatment of rheumatoid arthritis". Annals of the Rheumatic Diseases. 59 (11): 841–9. doi:10.1136/ard.59.11.841. PMC 1753034. PMID 11053058.
- Melchiorri D, Barbara vZ, Romaldas M, Nela V, Karsten BS, Ian H, Robert H, Harald E, Pierre D. "Assessment report. AUBAGIO (international non-proprietary name: teriflunomide). Procedure No. EMEA/H/C/002514/0000" (PDF). European Medicines Agency. European Medicines Agency. p. 119. Retrieved 5 June 2015.
- Rozman B (2002). "Clinical pharmacokinetics of leflunomide". Clinical Pharmacokinetics. 41 (6): 421–30. doi:10.2165/00003088-200241060-00003. PMID 12074690.
- "Clinical Pharmacology/Biopharmaceutics Review. Product: Arava (leflunomide tablets). Application Number: NDA 20905" (PDF). U.S. Food and Drug Administration (FDA). Retrieved 15 April 2016.
- "Summary of Opinion (Initial Authorisation): Aubagio (teriflunomide)" (PDF). European Medicines Agency. Retrieved 15 April 2016.
External links
- "Teriflunomide". Drug Information Portal. U.S. National Library of Medicine.