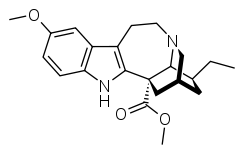
Voacangine
Voacangine (12-methoxyibogamine-18-carboxylic acid methyl ester) is an alkaloid found predominantly in the rootbark of the Voacanga africana tree, as well as in other plants such as Tabernanthe iboga, Tabernaemontana africana, Trachelospermum jasminoides, Tabernaemontana_divaricata and Ervatamia yunnanensis.[2][3][4][5] It is an iboga alkaloid which commonly serves as a precursor for the semi-synthesis of ibogaine.[6] It has also been demonstrated in animals to have similar anti-addictive properties to ibogaine itself.[7] It also potentiated effects of barbiturates[8]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
12-Methoxyibogamine-18-carboxylic acid, methyl ester | |
| Systematic IUPAC name
Methyl 17-ethyl-7-methoxy-3,13-diazapentacyclo[13.3.1.02,10.04,9.013,18] nonadeca-2(10),4,6,8-tetraene-1-carboxylate[1] | |
| Other names
Methyl 12-methoxyibogamine-18-carboxylate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.214.137 |
| MeSH | Voacangine |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C22H28N2O3 | |
| Molar mass | 368.477 g·mol−1 |
| Melting point | 136 to 137 °C (277 to 279 °F; 409 to 410 K) |
| log P | 3.748 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Side Effect
High doses of voacangine produces convulsions and asphyxia.[10]
gollark: Also not-very-expressiveness.
gollark: Unsafety, that's basically it.
gollark: But it's okay, and at least it's not C or something!
gollark: It's unfortunate that we got THIS as the way to develop basically everything for the interweb™.
gollark: More so than JS, arguably.
References
- "Compound Report Card CHEMBL182120 - Voacangine". ChEMBL.
- Patel, M. B.; Miet, C.; Poisson, J. (1967). "Alkaloids of some African Tabernaemontana". Annales Pharmaceutiques Françaises. 25 (5): 379–384. PMID 5611538.
- Fatima, T.; Ijaz, S.; Crank, G.; Wasti, S. (1987). "Indole Alkaloids from Trachelospermum jasminoides". Planta Medica. 53 (1): 57–59. doi:10.1055/s-2006-962620. PMID 17268963.
- Liu, G.; Liu, X.; Feng, X. Z. (1988). "Ervayunine: A New Indole Alkaloid from Ervatamia yunnanensis". Planta Medica. 54 (6): 519–521. doi:10.1055/s-2006-962535. PMID 3212080.
- Jenks, C. W. (2002). "Extraction Studies of Tabernanthe iboga and Voacanga africana". Natural Product Letters. 16 (1): 71–76. doi:10.1080/1057563029001/4881. PMID 11942686.
- US patent 2813873, "Derivatives of the Ibogaine Alkaloids", issued 1957-11-19
- Tsing Hua (January 28, 2006). "Antiaddictive Indole Alkaloids in Ervatamia yunnanensis and their Bioactivity". Academic Journal of Second Military Medical University.
- http://medind.nic.in/iby/t08/i4/ibyt08i4p317.pdf
- "Annals of the Brazilian Academy of Sciences" (PDF).
- https://www.erowid.org/plants/voacanga_africana/voacanga_africana_info1.shtml
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.