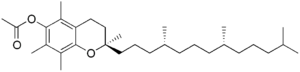
Tocopheryl acetate
α-Tocopheryl acetate, also known as vitamin E acetate, is a synthetic form of vitamin E. It is the ester of acetic acid and α-tocopherol.[2] The U.S. Centers for Disease Control and Prevention says that vitamin E acetate is a very strong culprit of concern in the 2019 outbreak of vaping-associated pulmonary injury (VAPI),[3] but there is not yet sufficient evidence to rule out contributions from other chemicals.[4]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
[(2R)-2,5,7,8-Tetramethyl-2-[(4R,8R)-4,8,12-trimethyltridecyl]chroman-6-yl] acetate | |
| Other names
α-Tocopherol acetate Vitamin E acetate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.369 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C31H52O3 | |
| Molar mass | 472.743 g/mol |
| Appearance | pale yellow, viscous liquid[1] |
| Melting point | –27.5 °C [1] |
| Boiling point | 240 °C decays without boiling[2] |
| insoluble[1] | |
| Solubility | soluble in acetone, chloroform, diethyl ether; poorly soluble in ethanol[1] |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Use in cosmetics
α-Tocopheryl acetate is often used in dermatological products such as skin creams. It is not oxidized and can penetrate through the skin to the living cells, where about 5% is converted to free tocopherol. Claims are made for beneficial antioxidant effects.[5] α-Tocopheryl acetate is used as an alternative to tocopherol itself because the phenolic hydroxyl group is blocked, providing a less acidic product with a longer shelf life. It is believed that the acetate is slowly hydrolyzed after it is absorbed into the skin, regenerating tocopherol and providing protection against the sun's ultraviolet rays.[6] Tocopheryl acetate was first synthesized in 1963 by workers at Hoffmann-La Roche.[7]
Although there is widespread use of tocopheryl acetate as a topical medication, with claims for improved wound healing and reduced scar tissue,[8] reviews have repeatedly concluded that there is insufficient evidence to support these claims.[9][10] There are reports of vitamin E-induced allergic contact dermatitis from use of vitamin E derivatives such as tocopheryl linoleate and tocopherol acetate in skin care products. Incidence is low despite widespread use.[11]
Misuse
Ingredient in vape liquids
On September 5, 2019, the United States Food and Drug Administration (US FDA) announced that 10 out of 18, or 56% of the samples of vape liquids sent in by states, linked to recent vaping related lung disease outbreak in the United States, tested positive for vitamin E acetate[12] which had been used as a thickening agent by illicit THC vape cartridge manufacturers.[13] On November 8, 2019, the Centers for Disease Control and Prevention (CDC) identified vitamin E acetate as a very strong culprit of concern in the vaping-related illnesses, but has not ruled out other chemicals or toxicants as possible causes.[3] The CDC's findings were based on fluid samples from the lungs of 29 patients with vaping-associated pulmonary injury, which provided direct evidence of vitamin E acetate at the primary site of injury in all the 29 lung fluid samples tested.[3] Research suggests when vitamin E acetate is inhaled, it may interfere with normal lung functioning.[4] A 2020 study found that vaporizing vitamin E acetate produced carcinogenic alkenes and benzene, but also exceptionally toxic ketene gas, which may be a contributing factor to the pulmonary injuries.[14]
Chemistry
At room temperature, α-tocopheryl acetate is a fat-soluble liquid. It has 3 chiral centers and thus 8 stereoisomers. It is made by esterifying α-tocopherol with acetic acid. 2R,4R,8R-isomer, also known as RRR-α-tocopheryl acetate, is the most common isomer used for various purposes. This is because α-tocopherol occurs in the nature primarily as RRR-α-tocopherol.[2]
α-Tocopherol acetate does not boil at atmospheric pressure and begins to degrade at 240 °C.[2] It can be vacuum distilled: it boils at 184 °C at 0.01 mmHg, at 194 °C (0.025 mmHg) and at 224 °C (0.3 mmHg). In practice, it is not degraded notably by air, visible light or UV-radiation. It has a refractive index of 1.4950–1.4972 at 20 °C.[1]
α-Tocopherol acetate is hydrolyzed to α-tocopherol and acetic acid under suitable conditions or when ingested by people.[2]
References
- The Merck index (12th ed.). Merck. 1996. p. 1580. ISBN 9780911910124.
- "Safety assessment of the substance α-tocopherol acetate for use in food contact materials". EFSA Journal. 14 (3): 4412. 2016. doi:10.2903/j.efsa.2016.4412.
- "Transcript of CDC Telebriefing: Update on Lung Injury Associated with E-cigarette Use, or Vaping". Centers for Disease Control and Prevention. 8 November 2019.

- "Outbreak of Lung Injury Associated with E-Cigarette Use, or Vaping". Centers for Disease Control and Prevention. 8 November 2019.

- Linus Pauling Institute Research Report: All About E at the Wayback Machine (archived 2015-02-23)
- Beijersbergen van Henegouwen G, Junginger H, de Vries H (1995). "Hydrolysis of RRR-alpha-tocopheryl acetate (vitamin E acetate) in the skin and its UV protecting activity (an in vivo study with the rat)". J Photochem Photobiol B. 29 (1): 45–51. doi:10.1016/1011-1344(95)90251-1. PMID 7472802.
- Mayer, H.; Schudel, P.; Rüegg, R.; Isler, O. (1963). "Über die Chemie des Vitamins E. 3. Mitteilung. Die Totalsynthese von (2R, 4′R, 8′R)- und (2S, 4′R, 8′R)-α-Tocopherol". Helvetica Chimica Acta. 46 (2): 650–671. doi:10.1002/hlca.19630460225. ISSN 0018-019X.
- Panin G, Strumia R, Ursini F (2004). "Topical alpha-tocopherol acetate in the bulk phase: eight years of experience in skin treatment". Ann. N. Y. Acad. Sci. 1031: 443–447. Bibcode:2004NYASA1031..443P. doi:10.1196/annals.1331.069. PMID 15753192.
- Sidgwick GP, McGeorge D, Bayat A (2015). "A comprehensive evidence-based review on the role of topicals and dressings in the management of skin scarring". Arch. Dermatol. Res. 307 (6): 461–477. doi:10.1007/s00403-015-1572-0. PMC 4506744. PMID 26044054.
- Tanaydin V, Conings J, Malyar M, van der Hulst R, van der Lei B (2016). "The Role of Topical Vitamin E in Scar Management: A Systematic Review". Aesthet Surg J. 36 (8): 959–965. doi:10.1093/asj/sjw046. PMID 26977069.
- Kosari P, Alikhan A, Sockolov M, Feldman SR (2010). "Vitamin E and allergic contact dermatitis". Dermatitis. 21 (3): 148–153. doi:10.2310/6620.2010.09083. PMID 20487657.
- Sun, Lena (September 6, 2019). "Contaminant found in marijuana vaping products linked to deadly lung illnesses, tests show". Washington Post. Retrieved 2019-09-09.
- "Three Companies Subpoenaed in Weed Vape Illness Investigation". Rolling Stone. September 10, 2019.
- Wu, D; O'Shea, DF (24 March 2020). "Potential for release of pulmonary toxic ketene from vaping pyrolysis of vitamin E acetate". Proceedings of the National Academy of Sciences of the United States of America. 117 (12): 6349–6355. doi:10.1073/pnas.1920925117. PMC 7104367. PMID 32156732.
