Supercritical water oxidation
Supercritical water oxidation (SCWO) is a process that occurs in water at temperatures and pressures above a mixture's thermodynamic critical point. Under these conditions water becomes a fluid with unique properties that can be used to advantage in the destruction of hazardous wastes such as PCBs. The fluid has a density between that of water vapor and liquid at standard conditions, and exhibits high gas-like diffusion rates along with high liquid-like collision rates. In addition, the behavior of water as a solvent is altered (in comparison to that of subcritical liquid water) - it behaves much less like a polar solvent. As a result, the solubility behavior is "reversed" so that chlorinated hydrocarbons become soluble in the water, allowing single-phase reaction of aqueous waste with a dissolved oxidizer. The reversed solubility also causes salts to precipitate out of solution, meaning they can be treated using conventional methods for solid-waste residuals. Efficient oxidation reactions occur at low temperature (400-650 °C) with reduced NOx production.
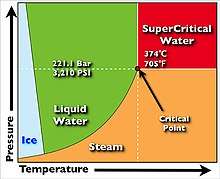
SCWO can be classified as green chemistry or as a Clean Technology. The elevated pressures and temperatures required for SCWO are routinely encountered in industrial applications such as petroleum refining and chemical synthesis.
A unique addition to the world of supercritical water (SCW) oxidation is generating high-pressure flames inside the SCW medium. The pioneer works on high-pressure Supercritical Water Flames were carried out by professor EU Franck at the German University of Karlsruhe in the late 80s. The works were mainly aimed at anticipating conditions which would cause spontaneous generation of non-desirable flames in the flameless SCW oxidation process. These flames would cause instabilities to the system and its components. ETH Zurich pursued the investigation of hydrothermal flames in continuously operated reactors. The rising needs for waste treatment and destruction methods motivated a Japanese Group in the Ebara Corporation to explore SCW flames as an environmental tool. Research on hydrothermal flames has also begun at NASA Glenn Research Center in Cleveland, Ohio.


Basic Research
Basic research on supercritical water oxidation was undertaken in the 1990s at Sandia National Laboratory's Combustion Research Facility (CRF), in Livermore, CA. Originally proposed as a hazardous waste destruction technology in response to the Kyoto protocol, multiple waste streams were studied by Steven F. Rice and Russ Hanush, and hydrothermal (supercritical water) flames were investigated by Richard R. Steeper and Jason D. Aiken. Among the waste streams studied were military dyes and pyrotechnics,[1][2] methanol,[3][4] and isopropyl alcohol.[5] Hydrogen peroxide was used as an oxidizing agent, and Eric Croiset was tasked with detailed measurements of the decomposition of hydrogen peroxide at supercritical water conditions.[6]
There were major engineering challenges associated with the deposition of salts[7] and chemical corrosion in these supercritical water reactors, and Anthony Lajeunesse led the team investigating these issues. To address these issues Lajeunesse designed a transpiring wall reactor[8] which introduced a pressure differential through the walls of an inner sleeve filled with pores to continuously rinse the inner walls of the reactor with fresh water. Russ Hanush was charged with the construction and operation of the supercritical fluids reactor (SFR)[9] used for these studies. Among its design intricacies were the inconel 625 alloy necessary for operation at such extreme temperatures and pressures, and the design of the high-pressure, high-temperature optical cells used for photometric access to the reacting flows which incorporated 24 carat gold pressure seals and sapphire windows.[10][11]
Commercial applications
Several companies in the United States are now working to commercialize supercritical reactors to destroy hazardous wastes. Widespread commercial application of SCWO technology requires a reactor design capable of resisting fouling and corrosion under supercritical conditions.
In Japan a number of commercial SCWO applications exist, among them one unit for treatment of halogenated waste built by Organo. In Korea two commercial size units have been built by Hanwha.
In Europe, Chematur Engineering AB of Sweden commercialized the SCWO technology for treatment of spent chemical catalysts to recover the precious metal, the AquaCat process. The unit has been built for Johnson Matthey in the UK. It is the only commercial SCWO unit in Europe and with its capacity of 3000 l/h it is the largest SCWO unit in the world. Chematur's Super Critical Fluids technology was acquired by SCFI Group (Cork, Ireland) who are actively commercializing the Aqua Critox SCWO process for treatment of sludge, e.g. de-inking sludge and sewage sludge. Many long duration trials on these applications have been made and thanks to the high destruction efficiency of 99.9%+ the solid residue after the SCWO process is well suited for recycling – in the case of de-inking sludge as paper filler and in the case of sewage sludge as phosphorus and coagulant. SCFI Group operate a 250 l/h Aqua Critox demonstration plant in Cork, Ireland.
Turbosystems Engineering (California, USA) is actively commercializing their patented transpiring wall SCWO reactor ("TWR") with a focus on renewable energy applications.
See also
References
- Rice, Steven (1994). "Supercritical Water Oxidation of Colored Smoke, Dye, and Pyrotechnic Compositions" (PDF). Physics Phor Phun.
- Rice, Steven (1995). "Destruction of Hazardous Munitions by Supercritical Water Oxidation" (PDF). Physics Phor Phun.
- Rice, Steven (1995). "Hydrothermal Oxidation Kinetics of Methanol" (PDF). Physics Phor Phun.
- Rice, Steven (1996). "Raman-Spectroscopic Measurement of Oxidation in Supercritical Water - Conversion of Methanol to Formaldehyde" (PDF). Physics Phor Phun.
- Hunter, Thomas (1996). "Raman-Spectroscopic Measurement of Oxidation in Supercritical Water - Conversion of Isopropanol to Acetone" (PDF). Physics Phor Phun.
- Croiset, Eric (1997). "Hydrogen Peroxide Decomposition in Supercritical Water" (PDF). Physics Phor Phun.
- Lajeunesse, Anthony (1993). "Salt Deposition Studies in a Supercritical Water Reactor" (PDF). Physics Phor Phun.
- Lajeunesse, Anthony (1997). "Kinetic Investigation of the Oxidation of Naval Excess Hazardous Materials in Supercritical Water for the Design of a Transpiration-Wall Reactor" (PDF). Physics Phor Phun.
- Hanush, Russ (1995). "Operation and Performance of the Supercritical Fluids Reactor" (PDF). Physics Phor Phun.
- Rice, Steven (1997). "Design Strategies for High-Temperature, High-Pressure Optical Cells" (PDF). Physics Phor Phun.
- Rice, Steven (2000). "Design strategies for Optically-Accessible High-Temperature, High-Pressure Reactor Cells" (PDF). Physics Phor Phun.
External links
There are some research groups working in this topic throughout the world:
- SCFI have a working AquaCritox A10 plant in Cork(Ireland)
- UVa High Pressure Processes Group (Spain)
- Clean Technology Group (UK)
- FZK Karlsruhe (Germany)
- ETH Zurich, Transport Processes and Reactions Laboratory (Switzerland)
- UCL (London, UK) Clean Materials technology group working on Continuous Hydrothermal Flow Synthesis
- UBC (Vancouver, BC) Mechanical Engineering Research Activities, including projects on SCWO
- Turbosystems Engineering SCWO technology
- Universidad de Cádiz (UCA). Supercritical Fluids Group
