Stryker's reagent
Stryker's reagent ([(PPh3)CuH]6),[1] also known as the Osborn complex, is a hexameric copper hydride ligated with triphenylphosphine. It is a brick red, air-sensitive solid. Stryker's reagent is a mildly hydridic reagent, used in homogeneous catalysis of conjugate reduction reactions of enones, enoates, and related substrates.
 | |
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ECHA InfoCard | 100.156.010 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C108H96Cu6P6 | |
| Molar mass | 1961.04 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation and structure

The compound is prepared by hydrogenation of copper(I) tert-butoxide, generated in situ from copper(I) chloride and sodium tert-butoxide.[2] Other more convenient methods have been developed since its discovery.[3][4]
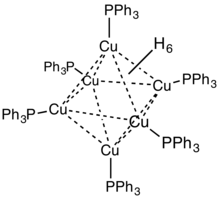
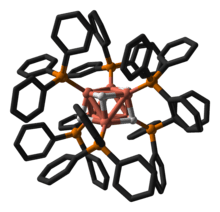
In terms of its structure, the compound is an octahedral cluster of Cu(PPh3) centres that are bonded by Cu---Cu and Cu---H interactions. Originally six of the eight faces were thought to be capped by hydride ligands.[5] This structural assignment was revised in 2014; the hydrides are now best described as edge bridging rather than face bridging.[6]
Applications in organic synthesis
The compound can effect regioselective conjugate reductions of various carbonyl derivatives including unsaturated aldehydes, ketones, and esters. This reagent was assigned as the "Reagent of the year" in 1991 for its functional group tolerance, high overall efficiency, and mild reaction conditions in the reduction reactions. Stryker's reagent is used in a catalytic amount where it is regenerated in the reaction in situ using a stoichiometric hydride source, often being molecular hydrogen or silanes. If stored under an inert atmosphere (e.g. argon, nitrogen) it has indefinite shelf life. Brief exposure to the oxygen does not destroy its activity significantly, although solvents used with Stryker's reagent should be rigorously degassed.[7]
Modifications to Stryker's reagent
Ligand-modified versions of Stryker's reagent have been reported. By changing the ligand to e.g. P(O-iPr)3 the selectivity can be improved significantly.[8] In addition, Lipshutz et. al., have shown that the addition of a bidentate, achiral bis-phosphine ligand on the Cu center can lead to substrate-to-ligand ratios typically on the order of 1000−10000:1 can be used to afford products in high yields.[9]
References
- Mahoney, W.S.; Brestensky, D.M.; Stryker, J.M. (1988). "Selective Hydride-mediated conjugate reduction of α,β-unsaturated Carbonyl Compounds Using [(Ph3P)CuH]6". J. Am. Chem. Soc. 110: 291–293. doi:10.1021/ja00209a048.
- Bezman, S. A.; Churchill, M. R.; Osborn, J. A.; Wormald, J. (1971). "Preparation and Crystallographic Characterization of a Hexameric Triphenylphosphinecopper Hydride Cluster". J. Am. Chem. Soc. 93 (8): 2063–2065. doi:10.1021/ja00737a045.
- O. Riant "Copper(I) hydride reagents and catalysts" Patai's Chemistry of Functional Groups, 2011, John Wiley & Sons. doi:10.1002/9780470682531.pat0448
- R. D. Stephens "Hydrido(Triphenylphosphine)Copper(I)" Inorganic Syntheses, 1979, vol. 19, pp. 87–89. doi:10.1002/9780470132500.ch17
- Raymond C. Stevens, Malcolm R. McLean, Robert Bau, Thomas F. Koetzle "Neutron diffraction structure analysis of a hexanuclear copper hydrido complex, H6Cu6[P(p-tolyl)3]6: an unexpected finding" J. Am. Chem. Soc. 1989, pp 3472–3473. doi:10.1021/ja00191a077
- Bennett, Elliot L.; Murphy, Patrick J.; Imberti, Silvia; Parker, Stewart F. (2014-03-17). "Characterization of the Hydrides in Stryker's Reagent: [HCu{P(C6H5)3}]6". Inorganic Chemistry. 53 (6): 2963–2967. doi:10.1021/ic402736t. ISSN 0020-1669. PMID 24571368.
- John F. Daeuble and Jeffrey M. Stryker "Hexa-μ-hydrohexakis(triphenylphosphine)hexacopper" eEROS Encyclopedia of Reagents for Organic Synthesis, 2001. doi:10.1002/047084289X.rh011m
- Andrejs Pelss, Esa T. T. Kumpulainen and Ari M. P. Koskinen J. Org. Chem. 2009, 74, 7598-7601. doi:10.1021/jo9017588
- Baker, Benjamin A.; Bošković, Žarko V.; Lipshutz, Bruce H. (2008). "(BDP)CuH: A "Hot" Stryker's Reagent for Use in Achiral Conjugate Reductions". Organic Letters. 10 (2): 289–292. doi:10.1021/ol702689v. PMID 18092793.
| Wikimedia Commons has media related to Stryker's reagent. |