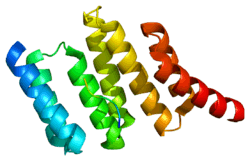Hop (protein)
Hop, occasionally written HOP, is an abbreviation for Hsp70-Hsp90 Organizing Protein. It functions as a co-chaperone which reversibly links together the protein chaperones Hsp70 and Hsp90.[5]
Hop belongs to the large group of co-chaperones, which regulate and assist the major chaperones (mainly heat shock proteins). It is one of the best studied co-chaperones of the Hsp70/Hsp90-complex. It was first discovered in yeast and homologues were identified in human, mouse, rat, insects, plants, parasites, and virus. The family of these proteins is referred to as STI1 (stress inducible protein) and can be divided into yeast, plant, and animal STI1 (Hop).
Synonyms
|
|
Gene
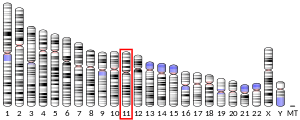
The gene for human Hop is located on chromosome 11q13.1 and consists of 14 exons.
Structure
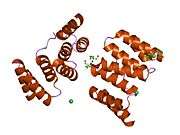
STI proteins are characterized by some structural features: All homologues have nine tetratricopeptide repeat (TPR) motifs, that are clustered into domains of three TPRs. The TPR motif is a very common structural feature used by many proteins and provides the ability of directing protein-protein interactions. Crystallographic structural information is available for the N-terminal TPR1 and the central TPR2A domains in complex with Hsp90 resp. Hsp70 ligand peptides.[6]
The Hsp70-Hsp90 Organizing Protein (Hop, STIP1 in humans) is the co-chaperone responsible for the transfer of client proteins between Hsp70 and Hsp90. Hop is evolutionarily conserved in Eukaryotes and is found in both the nucleus and cytoplasm[7]. Drosophila Hop is a monomeric protein that consists of three tetratricopeptide repeat domain regions (TPR1, TPR2A, TPR2B), one aspartic acid-proline repeat domain (DP). The TPR domains interact with the c-terminals of Hsp90 and Hsp70, with TPR1 and TPR2B binding to Hsp70 and TPR2A binding preferentially to Hsp90. The intermediate structures of heat shock machinery are difficult to characterize completely because of the transient and fast paced nature of chaperone function[8].
Function
The main function of Hop is to link Hsp70 and Hsp90 together. But recent investigations indicate that it also modulates the chaperone activities of the linked proteins and possibly interacts with other chaperones and proteins. Apart from its role in the Hsp70/Hsp90 "chaperone machine" it seems to participate in other protein complexes too (for example in the signal transduction complex EcR/USP and in the Hepatitis B virus reverse transcriptase complex, which enables the viral replication). It acts as a receptor for prion proteins too.[9][10] Hop is located in diverse cellular regions and also moves between the cytoplasm and the nucleus.
In Drosophila RNA interference pathways, Hop has been shown to be an integral part of the pre-RISC complex for siRNAs.[11] In the Drosophila Piwi-interacting RNA pathway, the RNA interference pathway responsible for the repression of transposable elements (transposons), Hop has been shown to interact with Piwi,[12] and in the absence of Hop, transposons are derepressed, leading to severe genomic instability and infertility.[13]
Interactions
Human Hop (STIP1) has been shown to interact with PRNP[14] and Heat shock protein 90kDa alpha (cytosolic), member A1.[15][16]
References
- GRCh38: Ensembl release 89: ENSG00000168439 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000024966 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Odunuga OO, Longshaw VM, Blatch GL (October 2004). "Hop: more than an Hsp70/Hsp90 adaptor protein". BioEssays. 26 (10): 1058–68. doi:10.1002/bies.20107. PMID 15382137.
- Scheufler C, Brinker A, Bourenkov G, Pegoraro S, Moroder L, Bartunik H, Hartl FU, Moarefi I (April 2000). "Structure of TPR domain-peptide complexes: critical elements in the assembly of the Hsp70-Hsp90 multichaperone machine". Cell. 101 (2): 199–210. doi:10.1016/S0092-8674(00)80830-2. PMID 10786835.
- Schmid AB, et al. (2012). "The architecture of functional modules in the Hsp90 co-chaperone Sti1/Hop". EMBO J. 31: 1506–17. doi:10.1038/emboj.2011.472. PMC 3321170. PMID 22227520.
- Yamamoto S, et al. (2014). "ATPase activity and ATP-dependent conformational change in the co-chaperone HSP70/HSP90-organizing protein (HOP)". J. Biol. Chem. 289: 9880–6. doi:10.1074/jbc.m114.553255. PMC 3975032. PMID 24535459.
- Martins VR, Graner E, Garcia-Abreu J, de Souza SJ, Mercadante AF, Veiga SS, Zanata SM, Neto VM, Brentani RR (December 1997). "Complementary hydropathy identifies a cellular prion protein receptor". Nature Medicine. 3 (12): 1376–82. doi:10.1038/nm1297-1376. PMID 9396608.
- Zanata SM, Lopes MH, Mercadante AF, Hajj GN, Chiarini LB, Nomizo R, Freitas AR, Cabral AL, Lee KS, Juliano MA, de Oliveira E, Jachieri SG, Burlingame A, Huang L, Linden R, Brentani RR, Martins VR (July 2002). "Stress-inducible protein 1 is a cell surface ligand for cellular prion that triggers neuroprotection". The EMBO Journal. 21 (13): 3307–16. doi:10.1093/emboj/cdf325. PMC 125391. PMID 12093732.
- Iwasaki S, Sasaki HM, Sakaguchi Y, Suzuki T, Tadakuma H, Tomari Y (May 2015). "Defining fundamental steps in the assembly of the Drosophila RNAi enzyme complex". Nature. 521 (7553): 533–6. doi:10.1038/nature14254. PMID 25822791.
- Gangaraju VK, Yin H, Weiner MM, Wang J, Huang XA, Lin H (February 2011). "Drosophila Piwi functions in Hsp90-mediated suppression of phenotypic variation". Nature Genetics. 43 (2): 153–8. doi:10.1038/ng.743. PMC 3443399. PMID 21186352.
- Karam JA, Parikh RY, Nayak D, Rosenkranz D, Gangaraju VK (April 2017). "Co-chaperone Hsp70/Hsp90-organizing protein (Hop) is required for transposon silencing and Piwi-interacting RNA (piRNA) biogenesis". The Journal of Biological Chemistry. 292 (15): 6039–6046. doi:10.1074/jbc.C117.777730. PMC 5391737. PMID 28193840.
- Zanata SM, Lopes MH, Mercadante AF, Hajj GN, Chiarini LB, Nomizo R, Freitas AR, Cabral AL, Lee KS, Juliano MA, de Oliveira E, Jachieri SG, Burlingame A, Huang L, Linden R, Brentani RR, Martins VR (July 2002). "Stress-inducible protein 1 is a cell surface ligand for cellular prion that triggers neuroprotection". The EMBO Journal. 21 (13): 3307–16. doi:10.1093/emboj/cdf325. PMC 125391. PMID 12093732.
- Scheufler C, Brinker A, Bourenkov G, Pegoraro S, Moroder L, Bartunik H, Hartl FU, Moarefi I (April 2000). "Structure of TPR domain-peptide complexes: critical elements in the assembly of the Hsp70-Hsp90 multichaperone machine". Cell. 101 (2): 199–210. doi:10.1016/S0092-8674(00)80830-2. PMID 10786835.
- Johnson BD, Schumacher RJ, Ross ED, Toft DO (February 1998). "Hop modulates Hsp70/Hsp90 interactions in protein folding". The Journal of Biological Chemistry. 273 (6): 3679–86. doi:10.1074/jbc.273.6.3679. PMID 9452498.
Further reading
- Rasmussen HH, van Damme J, Puype M, Gesser B, Celis JE, Vandekerckhove J (December 1992). "Microsequences of 145 proteins recorded in the two-dimensional gel protein database of normal human epidermal keratinocytes". Electrophoresis. 13 (12): 960–9. doi:10.1002/elps.11501301199. PMID 1286667.
- Honoré B, Leffers H, Madsen P, Rasmussen HH, Vandekerckhove J, Celis JE (April 1992). "Molecular cloning and expression of a transformation-sensitive human protein containing the TPR motif and sharing identity to the stress-inducible yeast protein STI1". The Journal of Biological Chemistry. 267 (12): 8485–91. PMID 1569099.
- Maruyama K, Sugano S (January 1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Bonaldo MF, Lennon G, Soares MB (September 1996). "Normalization and subtraction: two approaches to facilitate gene discovery". Genome Research. 6 (9): 791–806. doi:10.1101/gr.6.9.791. PMID 8889548.
- Dittmar KD, Pratt WB (May 1997). "Folding of the glucocorticoid receptor by the reconstituted Hsp90-based chaperone machinery. The initial hsp90.p60.hsp70-dependent step is sufficient for creating the steroid binding conformation". The Journal of Biological Chemistry. 272 (20): 13047–54. doi:10.1074/jbc.272.20.13047. PMID 9148915.
- Dittmar KD, Demady DR, Stancato LF, Krishna P, Pratt WB (August 1997). "Folding of the glucocorticoid receptor by the heat shock protein (hsp) 90-based chaperone machinery. The role of p23 is to stabilize receptor.hsp90 heterocomplexes formed by hsp90.p60.hsp70". The Journal of Biological Chemistry. 272 (34): 21213–20. doi:10.1074/jbc.272.34.21213. PMID 9261129.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (October 1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Zou J, Guo Y, Guettouche T, Smith DF, Voellmy R (August 1998). "Repression of heat shock transcription factor HSF1 activation by HSP90 (HSP90 complex) that forms a stress-sensitive complex with HSF1". Cell. 94 (4): 471–80. doi:10.1016/S0092-8674(00)81588-3. PMID 9727490.
- Scanlan MJ, Gordan JD, Williamson B, Stockert E, Bander NH, Jongeneel V, Gure AO, Jäger D, Jäger E, Knuth A, Chen YT, Old LJ (November 1999). "Antigens recognized by autologous antibody in patients with renal-cell carcinoma". International Journal of Cancer. 83 (4): 456–64. doi:10.1002/(SICI)1097-0215(19991112)83:4<456::AID-IJC4>3.0.CO;2-5. PMID 10508479.
- Scheufler C, Brinker A, Bourenkov G, Pegoraro S, Moroder L, Bartunik H, Hartl FU, Moarefi I (April 2000). "Structure of TPR domain-peptide complexes: critical elements in the assembly of the Hsp70-Hsp90 multichaperone machine". Cell. 101 (2): 199–210. doi:10.1016/S0092-8674(00)80830-2. PMID 10786835.
- Hernández MP, Chadli A, Toft DO (April 2002). "HSP40 binding is the first step in the HSP90 chaperoning pathway for the progesterone receptor". The Journal of Biological Chemistry. 277 (14): 11873–81. doi:10.1074/jbc.M111445200. PMID 11809754.
- Brinker A, Scheufler C, Von Der Mulbe F, Fleckenstein B, Herrmann C, Jung G, Moarefi I, Hartl FU (May 2002). "Ligand discrimination by TPR domains. Relevance and selectivity of EEVD-recognition in Hsp70 x Hop x Hsp90 complexes". The Journal of Biological Chemistry. 277 (22): 19265–75. doi:10.1074/jbc.M109002200. PMID 11877417.
- Zanata SM, Lopes MH, Mercadante AF, Hajj GN, Chiarini LB, Nomizo R, Freitas AR, Cabral AL, Lee KS, Juliano MA, de Oliveira E, Jachieri SG, Burlingame A, Huang L, Linden R, Brentani RR, Martins VR (July 2002). "Stress-inducible protein 1 is a cell surface ligand for cellular prion that triggers neuroprotection". The EMBO Journal. 21 (13): 3307–16. doi:10.1093/emboj/cdf325. PMC 125391. PMID 12093732.
- Hernández MP, Sullivan WP, Toft DO (October 2002). "The assembly and intermolecular properties of the hsp70-Hop-hsp90 molecular chaperone complex". The Journal of Biological Chemistry. 277 (41): 38294–304. doi:10.1074/jbc.M206566200. PMID 12161444.
- Abbas-Terki T, Briand PA, Donzé O, Picard D (September 2002). "The Hsp90 co-chaperones Cdc37 and Sti1 interact physically and genetically". Biological Chemistry. 383 (9): 1335–42. doi:10.1515/BC.2002.152. PMID 12437126.
- Imai Y, Soda M, Murakami T, Shoji M, Abe K, Takahashi R (December 2003). "A product of the human gene adjacent to parkin is a component of Lewy bodies and suppresses Pael receptor-induced cell death". The Journal of Biological Chemistry. 278 (51): 51901–10. doi:10.1074/jbc.M309655200. PMID 14532270.
- Longshaw VM, Chapple JP, Balda MS, Cheetham ME, Blatch GL (February 2004). "Nuclear translocation of the Hsp70/Hsp90 organizing protein mSTI1 is regulated by cell cycle kinases". Journal of Cell Science. 117 (Pt 5): 701–10. doi:10.1242/jcs.00905. PMID 14754904.
- Rush J, Moritz A, Lee KA, Guo A, Goss VL, Spek EJ, Zhang H, Zha XM, Polakiewicz RD, Comb MJ (January 2005). "Immunoaffinity profiling of tyrosine phosphorylation in cancer cells". Nature Biotechnology. 23 (1): 94–101. doi:10.1038/nbt1046. PMID 15592455.