Resminostat
Resminostat (4SC-201 or RAS2410) is an orally bioavailable inhibitor of histone deacetylases (HDACs), of which inhibitors are antineoplastic agents.[1]
In 2011, the German drug maker 4SC was granted orphan drug designation for resminostat by the US FDA for the treatment of hepatocellular carcinoma (HCC).[2]
 | |
| Names | |
|---|---|
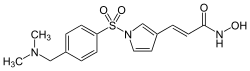
| IUPAC name
(2E)-3-[1-({4-[(Dimethylamino)methyl]phenyl}sulfonyl)-1H-pyrrol-3-yl]-N-hydroxyacrylamide | |
| Other names
RAS2410; Resminostat 4SC-201 | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C16H19N3O4S | |
| Molar mass | 349.41 g·mol−1 |
| Density | 1.282 g/cm3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
In 2016 the FDA granted [another?] IND for clinical tests in combination with sorafenib for HCC.[3] 4SC say "In several phase I and phase II trials, resminostat has already demonstrated very good safety and tolerability, alongside promising indications of efficacy."[3]
Clinical trials
It has undergone a phase I/II clinical trial for K-ras mutated advanced colorectal carcinoma.[4]
It has undergone a phase II clinical trial for relapsed or refractory Hodgkin's Lymphoma.[5]
Mechanism
Resminostat restrains the phosphorylation of 4E-BP1 and p70S6k, indicating an disturbance with Akt signalling pathway. The treatment of resminostat leads to a drop of Bim and Bax protein level and Bcl-xL level.[6]
As with other HDAC inhibitors such as pracinostat, the inhibition of HDACs by resminostat results in an accumulation of highly acetylated histones, followed by an abduction of chromatin remodeling, inhibition of tumor suppressor genes transcription and cell division, and finally tumor cell apoptosis.
References
- "Biological activity of resminostat in selleck chemicals research". selleck.
- "4SC's Anti-Cancer Drug Resminostat achieves Median Overall Survival of 8.0 Months in Second-Line Advanced Liver Cancer (HCC) Patients". September 2012.
- 4SC AG: FDA approves IND application for resminostat in liver cancer. Feb 2016
- 4SC-201 (Resminostat) in Advanced Colorectal Carcinoma (SHORE)
- Resminostat (4SC-201) in Relapsed or Refractory Hodgkin's Lymphoma (SAPHIRE)
- Brunetto AT, et al. (1 Oct 2013). "First-in-human, pharmacokinetic and pharmacodynamic phase I study of Resminostat, an oral histone deacetylase inhibitor, in patients with advanced solid tumors". Clin Cancer Res. 19 (19): 5494–504. doi:10.1158/1078-0432.CCR-13-0735. PMC 3790647. PMID 24065624.