Omaveloxolone
Omaveloxolone (RTA 408) is a second generation member of the synthetic oleanane triterpenoid compounds and currently in clinical development by Reata Pharmaceuticals. Preclinical studies have demonstrated that omaveloxolone possesses antioxidative and anti-inflammatory activities[1][2] and the ability to improve mitochondrial bioenergetics.[3] Omaveloxolone is currently under clinical investigation for a variety of indications, including Friedreich’s ataxia, mitochondrial myopathies, immunooncology, and prevention of corneal endothelial cell loss following cataract surgery.
 | |
| Clinical data | |
|---|---|
| Other names | RTA 408 |
| Routes of administration | Oral, Topical (Ophthalmic) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
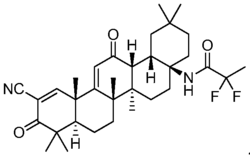
| Formula | C33H44F2N2O3 |
| Molar mass | 554.723 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Mechanism of action
The effects of omaveloxolone and related synthetic triterpenoid compounds have been documented in over 200 peer-reviewed scientific manuscripts.[4] The mechanism of action of omaveloxolone and its related compounds has been demonstrated to be through a combination of activation of the antioxidative transcription factor Nrf2 and inhibition of the pro-inflammatory transcription factor NF-κB.[1]
Nrf2 transcriptionally regulates multiple genes that play both direct and indirect roles in producing antioxidative potential and the production of cellular energy (i.e., adenosine triphosphate or ATP) within the mitochondria. Consequently, unlike exogenously administered antioxidants (e.g., vitamin E or Coenzyme Q10), which provide a specific and finite antioxidative potential, omaveloxolone, through Nrf2, broadly activates intracellular and mitochondrial antioxidative pathways, in addition to pathways that may directly increase mitochondrial biogenesis (such as PGC1α) and bioenergetics.[3]
Research
Friedreich's Ataxia
There appears to be dose-dependent induction of Nrf2 target genes, along with induction of biomarkers of mitochondrial function in one early clinical trial.[5] Time- and dose-dependent neurological improvements were observed.[5] A phase 1 with patients was completed in 2017 and results from the phase 2 trail where showed on October 2019.[6]
References
- Reisman SA, Lee CY, Meyer CJ, Proksch JW, Ward KW (Jul 2014). "Topical application of the synthetic triterpenoid RTA 408 activates Nrf2 and induces cytoprotective genes in rat skin". Archives of Dermatological Research. 306 (5): 447–54. doi:10.1007/s00403-013-1433-7. PMID 24362512.
- Reisman SA, Lee CY, Meyer CJ, Proksch JW, Sonis ST, Ward KW (May 2014). "Topical application of the synthetic triterpenoid RTA 408 protects mice from radiation-induced dermatitis". Radiation Research. 181 (5): 512–20. Bibcode:2014RadR..181..512R. doi:10.1667/RR13578.1. PMID 24720753.
- Neymotin A, Calingasan NY, Wille E, Naseri N, Petri S, Damiano M, Liby KT, Risingsong R, Sporn M, Beal MF, Kiaei M (Jul 2011). "Neuroprotective effect of Nrf2/ARE activators, CDDO ethylamide and CDDO trifluoroethylamide, in a mouse model of amyotrophic lateral sclerosis". Free Radical Biology & Medicine. 51 (1): 88–96. doi:10.1016/j.freeradbiomed.2011.03.027. PMC 3109235. PMID 21457778.
- "Pubmed Search". April 15, 2015. Retrieved April 15, 2015.
- Strawser, Cassandra; Schadt, Kimberly; Hauser, Lauren; McCormick, Ashley; Wells, McKenzie; Larkindale, Jane; Lin, Hong; Lynch, David R. (2017-07-26). "Pharmacological therapeutics in Friedreich ataxia: the present state". Expert Review of Neurotherapeutics. 17 (9): 895–907. doi:10.1080/14737175.2017.1356721. ISSN 1744-8360. PMID 28724340.
- "Reata Announces Positive Topline Results from the MOXIe Registrational Trial of Omaveloxolone in Patients with Friedreich's Ataxia".