Newman–Kwart rearrangement
The Newman–Kwart rearrangement is a type of rearrangement reaction in which the aryl group of an O-aryl thiocarbamate, ArOC(=S)NMe2, migrates from the oxygen atom to the sulfur atom, forming an S-aryl thiocarbamate, ArSC(=O)NMe2.[1][2][3] The reaction is named after its discoverers, Melvin Spencer Newman[4] and Harold Kwart.[5]
| Newman-Kwart rearrangement | |
|---|---|
| Named after | Melvin Spencer Newman Harold Kwart |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| Organic Chemistry Portal | newman-kwart-rearrangement |
| RSC ontology ID | RXNO:0000412 |

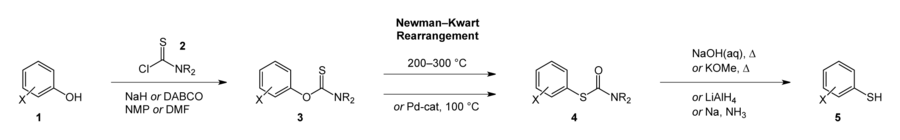
The Newman–Kwart rearrangement is one of the most important ways of synthesising thiophenols. A phenol (1) is deprotonated with a base such as DABCO or NaH and reacted with a thiocarbamoyl chloride (2) to form an O-aryl thiocarbamate (3). Heating 3 to around 250 °C causes it undergo Newman–Kwart rearrangement to an S-aryl thiocarbamate (4). Alkaline hydrolysis or similar cleavage yields a thiophenol (5).[6][7]
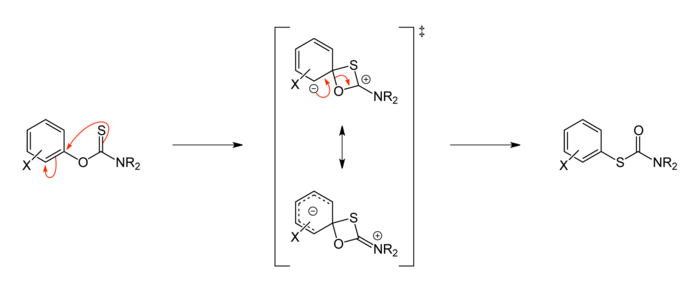
Mechanism
The Newman–Kwart rearrangement is intramolecular; it proceeds via a four-membered cyclic transition state.[3][6]
Real World Application
- Isomazole
See also
- Smiles rearrangement
- Chapman rearrangement
References
- Zonta, C.; De Lucchi, O.; Volpicelli, R.; Cotarca, L. (2007). Thione–Thiol Rearrangement: Miyazaki–Newman–Kwart Rearrangement and Others. Topics in Current Chemistry. 275. Springer Berlin / Heidelberg. pp. 131–161. doi:10.1007/128_065. ISBN 978-3-540-68099-4. ISSN 0340-1022.
- Harvey, J. N.; Jover, J.; Lloyd-Jones, G. C.; Moseley, J. D.; Murray, P. M.; Renny, J. S. (2009). "The Newman–Kwart Rearrangement of O-Aryl Thiocarbamates: Substantial Reduction in Reaction Temperatures through Palladium Catalysis". Angew. Chem. Int. Ed. 48: 7612–7615. doi:10.1002/anie.200903908. PMID 19746383.
- Lloyd-Jones, G. C.; Moseley, J. D.; Renny, J. S. (2008). "Mechanism and Application of the Newman-Kwart O→S Rearrangement of O-Aryl Thiocarbamates". Synthesis. 2008 (5): 661–689. doi:10.1055/s-2008-1032179.
- Newman, M. S.; Karnes, H. A. (1966). "The Conversion of Phenols to Thiophenols via Dialkylthiocarbamates". J. Org. Chem. 31 (12): 3980–3984. doi:10.1021/jo01350a023.
- Kwart, H.; Evans, E. R. (1966). "The Vapor Phase Rearrangement of Thioncarbonates and Thioncarbamates". J. Org. Chem. 31 (2): 410–413. doi:10.1021/jo01340a015.
- Burns, Matthew; Lloyd-Jones, G. C.; Moseley, J. D.; Renny, J. S. (2010). "The Molecularity of the Newman−Kwart Rearrangement". J. Org. Chem. 75 (19): 6347–6353. doi:10.1021/jo1014382.
- Melvin S. Newman, Frederick W. Hetzel (1971). "Thiophenols from Phenols: 2-Naphthalenethiol". Org. Synth. 51: 139. doi:10.15227/orgsyn.051.0139.CS1 maint: uses authors parameter (link)