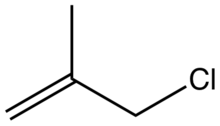
Methallyl chloride
Methallyl chloride is the organic compound with the formula CH2=C(CH3)CH2Cl. It is a colorless liquid and a lacrymator. Its properties are similar to those of allyl chloride. It is a strong alkylating agent used to install isobutenyl groups.[1]
 | |
| Names | |
|---|---|
| IUPAC name
3-Chloro-2-methyl-1-propene | |
| Other names
Isobutenyl chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.008.411 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2554 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H7Cl | |
| Molar mass | 90.55 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.9210 g/cm3 (15 °C) |
| Boiling point | 160–162 °C (320–324 °F; 433–435 K) |
| Hazards | |
| GHS pictograms |       |
| GHS Signal word | Danger |
GHS hazard statements |
H225, H302, H314, H317, H331, H335, H336, H351, H361, H372, H373, H401, H411 |
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P272, P273, P280, P281, P301+312, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P308+313, P310 | |
| Flash point | −12 °C (10 °F; 261 K) |
| 540 °C (1,004 °F; 813 K) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Reactivity
It is also a precursor to methallyl ligand. It is an isomer of crotyl chloride.
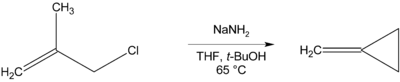
Methylenecyclopropane can be synthesised via an intramolecular cyclisation reaction from methallyl chloride by treatment with a strong base such as sodium amide.[2]
gollark: Oh, and you have to write it all in JS.
gollark: It's like normal sane server rendering but stupider because it sends along the JS bundle *too*.
gollark: Of course, like all intellectual people I run my web hosting on the microcontroller doing the onscreen display for my monitor.
gollark: baidicoot was working on some simple breadboard computer, it seems to be going slowly.
gollark: Ah.
References
- Krook, Mark A. (2007). "Methallyl chloride". e-EROS Encyclopedia of Reagents for Organic Synthesis. pp. 1–6. doi:10.1002/9780470842898.rm061.pub2.CS1 maint: uses authors parameter (link)
- Salaun, J. R.; Champion, J.; Conia, J. M. (1977). "Cyclobutanone from Methylenecyclopropane via Oxaspiropentane". 57: 36. doi:10.15227/orgsyn.057.0036. Cite journal requires
|journal=(help)
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.