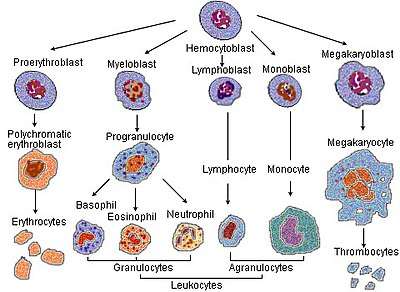
Erythropoiesis
Erythropoiesis (from Greek 'erythro' meaning "red" and 'poiesis' meaning "to make") is the process which produces red blood cells (erythrocytes), which is the development from erythropoietic stem cell to mature red blood cell.[2]
It is stimulated by decreased O2 in circulation, which is detected by the kidneys, which then secrete the hormone erythropoietin.[3] This hormone stimulates proliferation and differentiation of red cell precursors, which activates increased erythropoiesis in the hemopoietic tissues, ultimately producing red blood cells (erythrocytes).[3] In postnatal birds and mammals (including humans), this usually occurs within the red bone marrow.[3] In the early fetus, erythropoiesis takes place in the mesodermal cells of the yolk sac. By the third or fourth month, erythropoiesis moves to the liver.[4] After seven months, erythropoiesis occurs in the bone marrow. Increased level of physical activity can cause an increase in erythropoiesis.[5] However, in humans with certain diseases and in some animals, erythropoiesis also occurs outside the bone marrow, within the spleen or liver. This is termed extramedullary erythropoiesis.
The bone marrow of essentially all the bones produces red blood cells until a person is around five years old. The tibia and femur cease to be important sites of hematopoiesis by about age 25; the vertebrae, sternum, pelvis and ribs, and cranial bones continue to produce red blood cells throughout life. Up to the age of 20 years RBCs are produced from red bone marrow of all the bones (long bones and all the flat bones). After the age of 20 years, RBCs are produced from membranous bones such as vertebrae, the sternum, ribs, scapulas, and the iliac bones. After 20 years of age, the shaft of the long bones becomes yellow bone marrow because of fat deposition and loses the erythropoietic function.
Erythrocyte differentiation
In the process of red blood corpuscle maturation, a cell undergoes a series of differentiations. The following stages of development all occur within the bone marrow:
- A hemocytoblast, a multipotent hematopoietic stem cell, becomes
- a common myeloid progenitor or a multipotent stem cell, and then
- a unipotent stem cell, then
- a pronormoblast, also commonly called an proerythroblast or a rubriblast.
- This becomes a basophilic or early normoblast, also commonly called an erythroblast, then
- a polychromatophilic or intermediate normoblast, then
- an orthochromatic or late normoblast. At this stage the nucleus is expelled before the cell becomes
- a reticulocyte.
The cell is released from the bone marrow after Stage 7, and so in newly circulating red blood cells there are about 1% reticulocytes. After one to two days, these ultimately become "erythrocytes" or mature red blood cells.
These stages correspond to specific appearances of the cell when stained with Wright's stain and examined by light microscopy, and correspond to other biochemical changes.
In the process of maturation, a basophilic pronormoblast is converted from a cell with a large nucleus and a volume of 900 fL to an enucleated disc with a volume of 95 fL. By the reticulocyte stage, the cell has extruded its nucleus, but is still capable of producing hemoglobin.
Essential for the maturation of red blood cells are Vitamin B12 (cobalamin) and Vitamin B9 (Folic acid). Lack of either causes maturation failure in the process of erythropoiesis, which manifests clinically as reticulocytopenia, an abnormally low amount of reticulocytes.
Characteristics seen in erythrocytes during erythropoiesis
As they mature, a number of erythrocyte characteristics change: The overall size of the erythroid precursor cell reduces with the cytoplasmic to nucleus (C:N) ratio increasing. The nuclear diameter decreases and chromatin condenses with the staining reaction progressing from purplish red to dark blue at the final nuclear stage of Orthochromatic erythroblast, prior to nuclear ejection. The colour of the cytoplasm changes from blue at proerthroblast and basophilic stages to a pinkish red as a result of the increasing expression of haemoglobin as the cell develops. Initially, the nucleus is large in size and contains open chromatin. But, as red blood cells mature, the size of the nucleus decreases, until it finally disappears with the condensation of the chromatin material.[6]
Regulation of erythropoiesis
A feedback loop involving erythropoietin helps regulate the process of erythropoiesis so that, in non-disease states, the production of red blood cells is equal to the destruction of red blood cells and the red blood cell number is sufficient to sustain adequate tissue oxygen levels but not so high as to cause sludging, thrombosis, or stroke. Erythropoietin is produced in the kidney and liver in response to low oxygen levels. In addition, erythropoietin is bound by circulating red blood cells; low circulating numbers lead to a relatively high level of unbound erythropoietin, which stimulates production in the bone marrow.
Recent studies have also shown that the peptide hormone hepcidin may play a role in the regulation of hemoglobin production, and thus affect erythropoiesis. The liver produces hepcidin. Hepcidin controls iron absorption in the gastrointestinal tract and iron release from reticuloendothelial tissue. Iron must be released from macrophages in the bone marrow to be incorporated into the heme group of hemoglobin in erythrocytes. There are colony forming units that the cells follow during their formation. These cells are referred to as the committed cells including the granulocyte monocyte colony forming units.
The secretion of hepcidin is inhibited by another hormone, erythroferrone, produced by erythroblasts in response to erythropoietin, and identified in 2014.[7][8] It appears that this links erythropoietin-driven eyrthropoiesis with the iron mobilization needed for hemoglobin synthesis.
Loss of function of the erythropoietin receptor or JAK2 in mice cells causes failure in erythropoiesis, so production of red blood cells in embryos and growth is disrupted. If there is no systemic feedback inhibition, for example, the diminishment or absence of suppressors of cytokine signaling proteins, giantism may result as shown in mice models.[9][10]
See also
- Anemia: a condition with an abnormally low level of functional haemoglobin
- Polycythemia: a condition with an abnormally high level of red blood cells
- Dyserythropoiesis: a problem with the development of red blood cells
References
- Le, Tao; Bhushan, Vikas; Vasan, Neil (2010). First Aid for the USMLE Step 1: 2010 20th Anniversary Edition. USA: The McGraw-Hill Companies, Inc. pp. 123. ISBN 978-0-07-163340-6.
- Pelley, John W. (2007-01-01). "Amino Acid and Heme Metabolism". Elsevier's Integrated Biochemistry. ScienceDirect. pp. 97–105. doi:10.1016/B978-0-323-03410-4.50018-3. ISBN 9780323034104.
Erythropoiesis
Heme synthesis is coordinated with globin synthesis during erythropoiesis and as such does not occur in the mature erythrocyte. Erythropoiesis is the development of mature red blood cells from erythropoietic stem cells. The first cell that is morphologically recognizable in the red cell pathway is the proerythroblast. In the basophilic erythroblast, the nucleus becomes somewhat smaller, exhibiting a coarser appearance, and the cytoplasm becomes more basophilic owing to the presence of ribosomes. As the cell begins to produce hemoglobin, the cytoplasm attracts both basic and eosin stains and is called a polychromatophilic erythroblast. As maturation continues, the orthochromatophilic erythroblast extrudes its nucleus and the cell enters the circulation as a reticulocyte. As reticulocytes lose their polyribosomes, they become mature red blood cells. - Sherwood, L, Klansman, H, Yancey, P: Animal Physiology, Brooks/Cole, Cengage Learning, 2005.
- Palis J, Segel GB (June 1998). "Developmental biology of erythropoiesis". Blood Rev. 12 (2): 106–14. doi:10.1016/S0268-960X(98)90022-4. PMID 9661799.
- Le, Tao; Bhushan, Vikas; Vasan, Neil (2010). First Aid for the USMLE Step 1: 2010 20th Anniversary Edition. USA: The McGraw-Hill Companies, Inc. pp. 124. ISBN 978-0-07-163340-6.
- Textbook of Physiology by Dr. A. K. Jain reprint 2006-2007 3rd edition.
- Koury, M.J. (2015-01-13). "Erythroferrone: A Missing Link in Iron Regulation". The Hematologist. American Society of Hematology. Retrieved 26 August 2015.
- Kautz L, Jung G, Valore EV, Rivella S, Nemeth E, Ganz T (Jul 2014). "Identification of erythroferrone as an erythroid regulator of iron metabolism". Nature Genetics. 46 (7): 678–84. doi:10.1038/ng.2996. PMC 4104984. PMID 24880340.
- Nicolas G, Bennoun M, Porteu A, Mativet S, Beaumont C, Grandchamp B, Sirito M, Sawadogo M, Kahn A, Vaulont S (April 2002). "Severe iron deficiency anemia in transgenic mice expressing liver hepcidin". Proc. Natl. Acad. Sci. U.S.A. 99 (7): 4596–601. Bibcode:2002PNAS...99.4596N. doi:10.1073/pnas.072632499. PMC 123693. PMID 11930010.
- Michael Föller; Stephan M. Huber; Florian Lang (August 2008). "Erythrocyte programmed cell death". IUBMB Life. 60 (10): 661–668. doi:10.1002/iub.106. PMID 18720418.
Chinedu Ezeonyido