Alginic acid
Alginic acid, also called algin, is a polysaccharide distributed widely in the cell walls of brown algae that is hydrophilic and forms a viscous gum when hydrated. With metals such as sodium and calcium, its salts are known as alginates. It is a significant component of the biofilms produced by the bacterium Pseudomonas aeruginosa, a major pathogen found in the lungs of some people who have cystic fibrosis.[1] The biofilm and P. aeruginosa have a high resistance to antibiotics,[2] and are susceptible to inhibition by macrophages.[3] Its colour ranges from white to yellowish-brown. It is sold in filamentous, granular, or powdered forms.
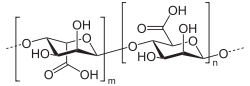 | |
| Names | |
|---|---|
| Other names
Alginic acid; E400; [D-ManA(β1→4)L-GulA(α1→4)]n | |
| Identifiers | |
| ChemSpider |
|
| ECHA InfoCard | 100.029.697 |
| EC Number |
|
| E number | E400 (thickeners, ...) |
| UNII | |
CompTox Dashboard (EPA) |
|
| Properties | |
| (C6H8O6)n | |
| Molar mass | 10,000 – 600,000 |
| Appearance | white to yellow, fibrous powder |
| Density | 1.601 g/cm3 |
| Acidity (pKa) | 1.5–3.5 |
| Pharmacology | |
| A02BX13 (WHO) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |

Structure
Alginic acid is a linear copolymer with homopolymeric blocks of (1-4)-linked β-D-mannuronate (M) and its C-5 epimer α-L-guluronate (G) residues, respectively, covalently linked together in different sequences or blocks. The monomers may appear in homopolymeric blocks of consecutive G-residues (G-blocks), consecutive M-residues (M-blocks) or alternating M and G-residues (MG-blocks).
Forms
Alginates are refined from brown seaweeds. Throughout the world, many of the Phaeophyceae class brown seaweeds are harvested to be processed and converted into sodium alginate. Sodium alginate is used in many industries including food, animal food, fertilisers, textile printing, and pharmaceuticals. Dental impression material uses alginate as its means of gelling. Food grade alginate an approved ingredient in process and manufactured foods.[4]
Brown seaweeds range in size from the giant kelp Macrocystis pyrifera which can be 20-40 meters long, to thick, leather-like seaweeds from 2–4 m long, to smaller species 30–60 cm long. Most brown seaweed used for alginates are gathered from the wild, with the exception of Laminaria japonica, which is cultivated in China for food and its surplus material is diverted to the alginate industry in China.
Alginates from different species of brown seaweed vary in their chemical structure resulting in different physical properties of alginates. Some species yield an alginate that gives a strong gel, another a weaker gel, some may produce a cream or white alginate, while others are difficult to gel and are best used for technical applications where color does not matter.[5]
Commercial grade alginate are extracted from giant kelp Macrocystis pyrifera, Ascophyllum nodosum, and types of Laminaria. Alginates are also is produced by two bacterial genera Pseudomonas and Azotobacter, which played a major role in the unravelling of its biosynthesis pathway. Bacterial alginates are useful for the production of micro- or nanostructures suitable for medical applications.[6]
Sodium alginate (NaC6H7O6) is the sodium salt of alginic acid. Sodium alginate is a gum.
Potassium alginate (KC6H7O6 ) is the potassium salt of alginic acid.
Calcium alginate (C12H14CaO12 ), is made from sodium alginate from which the sodium ion has been removed and replaced with calcium.
Production
The manufacturing process used to extract sodium alginates from brown seaweed fall into two categories: 1) Calcium alginate method and, 2) Alginic acid method.
Chemically the process is simple, but difficulties arise from the physical separations required between the slimy residues from viscous solutions and the separation of gelatinous precipitates that hold large amounts of liquid within the structure so they resist filtration and centrifugation.[7]
Uses
Alginate absorbs water quickly, which makes it useful as an additive in dehydrated products such as slimming aids, and in the manufacture of paper and textiles. It also is used for waterproofing and fireproofing fabrics, in the food industry as a thickening agent for drinks, ice cream, cosmetics, and as a gelling agent for jellies.
Alginate is used as an ingredient in various pharmaceutical preparations, such as Gaviscon, in which it combines with bicarbonate to inhibit reflux. Sodium alginate is used as an impression-making material in dentistry, prosthetics, lifecasting, and for creating positives for small-scale casting.
Sodium alginate is used in reactive dye printing and as a thickener for reactive dyes in textile screen-printing. Alginates do not react with these dyes and wash out easily, unlike starch-based thickeners.
As a material for micro-encapsulation.[8]
Calcium alginate is used in different types of medical products, including skin wound dressings to promote healing,[9][10] and may be removed with less pain than conventional dressings.
Alginate hydrogels
Alginate may be used in a hydrogel consisting of microparticles or bulk gels combined with nerve growth factor in bioengineering research to stimulate brain tissue for possible regeneration.[11] In research on bone reconstruction, alginate composites have favorable properties encouraging regeneration, such as improved porosity, cell proliferation, and mechanical strength, among other factors.[12]
See also
- Hyaluronic acid: a polysaccharide in animals.
- Agar
References
- Davies, JC (2002). "Pseudomonas aeruginosa in cystic fibrosis: pathogenesis and persistence". Paediatric Respiratory Reviews. 3 (2): 128–34. doi:10.1016/S1526-0550(02)00003-3. ISSN 1526-0542. PMID 12297059.
- Boyd, A; Chakrabarty, AM (1995). "Pseudomonas aeruginosa biofilms: role of the alginate exopolysaccharide". Journal of Industrial Microbiology. 15 (3): 162–8. doi:10.1007/BF01569821. ISSN 0169-4146. PMID 8519473.
- Leid, JG; Willson, CJ; Shirtliff, ME; Hassett, DJ; Parsek, MR; Jeffers, AK (1 November 2005). "The exopolysaccharide alginate protects Pseudomonas aeruginosa biofilm bacteria from IFN-gamma-mediated macrophage killing" (PDF). Journal of Immunology. 175 (11): 7512–8. doi:10.4049/jimmunol.175.11.7512. ISSN 0022-1767. PMID 16301659.
- "Alginates" (PDF). Agricultural Marketing Service, US Department of Agriculture. 5 February 2015. Retrieved 1 March 2018.
- FAO FISHERIES TECHNICAL PAPER 441, Tevita Bainiloga Jnr, School of Chemistry, University College, University of New South Wales and Australian Defence Force Academy Canberra Australia
- Remminghorst and Rehm (2009). "Microbial Production of Alginate: Biosynthesis and Applications". Microbial Production of Biopolymers and Polymer Precursors. Caister Academic Press. ISBN 978-1-904455-36-3.
- FAO Fisheries Technical Paper, 2003
- Aizpurua-Olaizola, Oier; Navarro, Patricia; Vallejo, Asier; Olivares, Maitane; Etxebarria, Nestor; Usobiaga, Aresatz (2016-01-01). "Microencapsulation and storage stability of polyphenols from Vitis vinifera grape wastes". Food Chemistry. 190: 614–621. doi:10.1016/j.foodchem.2015.05.117. PMID 26213018.
- Lansdown AB (2002). "Calcium: a potential central regulator in wound healing in the skin". Wound Repair Regen. 10 (5): 271–85. doi:10.1046/j.1524-475x.2002.10502.x. PMID 12406163.
- Stubbe, Birgit; Mignon, Arn; Declercq, Heidi; Vlierberghe, Sandra Van; Dubruel, Peter (2019). "Development of Gelatin-Alginate Hydrogels for Burn Wound Treatment". Macromolecular Bioscience. 19 (8): 1900123. doi:10.1002/mabi.201900123. ISSN 1616-5195. PMID 31237746.
- Büyüköz, M.; Erdal, E.; Altinkaya, S.A. (2016). "Nanofibrous gelatin scaffolds integrated with NGF-loaded alginate microspheres for brain tissue engineering". J. Tissue Eng. Regen. Med. 12 (2): e707–e719. doi:10.1002/term.2353. PMID 27863118. S2CID 206528926.
- Venkatesan, J; Bhatnagar, I; Manivasagan, P; Kang, K. H.; Kim, S. K. (2015). "Alginate composites for bone tissue engineering: A review". International Journal of Biological Macromolecules. 72: 269–81. doi:10.1016/j.ijbiomac.2014.07.008. PMID 25020082.
External links
- Alginate seaweed sources
- Alginate properties
- Alginate medical uses
- article Wired on Easy Cheese, describing sodium alginate