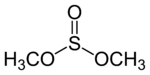
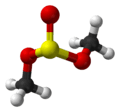
Dimethyl sulfite
Dimethyl sulfite is a sulfite ester with the chemical formula (CH3O)2SO.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Methoxysulfinyloxymethane | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.009.529 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C2H6O3S | |||
| Molar mass | 110.13 g·mol−1 | ||
| Appearance | Clear liquid | ||
| Density | 1.29 g/cm3 | ||
| Boiling point | 126 °C (259 °F; 399 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Dimethyl sulfite is used as an additive in some polymers to prevent oxidation.[2] It is also a potentially useful high energy battery electrolyte solvent.[3]
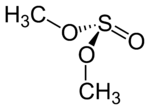
Structure and conformation
The dimethyl sulfite molecule can adopt several conformations. The most stable is the GG conformer.[1] Each C–O bond is gauche to the S=O bond, depicted below.
Preparation
Dimethyl sulfite is prepared from a 1:2 ratio of thionyl chloride and methanol.[4] The reaction can be catalyzed by tertiary amine bases and likely proceeds via the chlorosulfinate (MeOS(O)Cl),[5] this intermediate will exist only fleetingly in the presence of methanol and as such its decomposition to methyl chloride and sulfur dioxide (via the slower SNi mechanism) is not observed to any great extent.
- SOCl
2 + 2 CH
3OH → (CH
3O)
2SO + 2 HCl
See also
- Methyl methanesulfonate, a chemical with the same molecular formula but different arrangement of atoms
- Diethyl sulfite, a similar sulfite ester
- Dimethyl sulfoxide
- Dimethyl sulfate, a sulfate ester
References
- Borba, A.; Gómez-Zavaglia, A.; Simões, P. N. N. L.; Fausto, R. (2005). "Matrix Isolation FTIR Spectroscopic and Theoretical Study of Dimethyl Sulfite" (PDF). J. Phys. Chem. A. 109 (16): 3578–3586. doi:10.1021/jp050020t. hdl:10316/12879. PMID 16839024.
- Guenther, A.; Koenig, T.; Habicher, W. D.; Schwetlick, K. (1997). "Antioxidant action of organic sulfites. I. Esters of sulfurous acid as secondary antioxidants". Polymer Degradation and Stability. 55 (2): 209–216. doi:10.1016/S0141-3910(96)00150-4.
- N. P. Yao; E. D'Orsay; D. N. Bennion (1968). "Behavior of Dimethyl Sulfite as a Potential Nonaqueous Battery Solvent". J. Electrochem. Soc. 115 (10): 999–1003. doi:10.1149/1.2410917.
- Voss, Walter; Blanke, Erich (1931). ".Über die Ester der schwefligen Säure (Esters of sulfurous acid)". Justus Liebigs Annalen der Chemie. 485: 258–83. doi:10.1002/jlac.19314850116.
- van Woerden, H. F. (December 1963). "Organic Sulfites". Chemical Reviews. 63 (6): 557–571. doi:10.1021/cr60226a001.