Beta thymosins
Beta thymosins are a family of proteins which have in common a sequence of about 40 amino acids similar to the small protein thymosin β4. They are found almost exclusively in multicellular animals. Thymosin β4 was originally obtained from the thymus in company with several other small proteins which although named collectively "thymosins" are now known to be structurally and genetically unrelated and present in many different animal tissues.
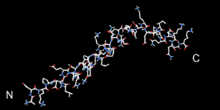
Single domain β-thymosins
Distribution
| Thymosin beta-4 family | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
 Structure of thymosin beta 9.[3] | |||||||||||
| Identifiers | |||||||||||
| Symbol | Thymosin | ||||||||||
| Pfam | PF01290 | ||||||||||
| InterPro | IPR001152 | ||||||||||
| SMART | SM00152 | ||||||||||
| PROSITE | PDOC00433 | ||||||||||
| SCOPe | 1hj0 / SUPFAM | ||||||||||
| |||||||||||
Monomeric β-thymosins, i.e. those of molecular weight similar to the peptides originally isolated from thymus by Goldstein, are found almost exclusively in cells of multicellular animals.[4] Known exceptions are monomeric thymosins found in a few single-celled organisms, significantly those currently regarded as the closest relatives of multicellular animals:[5] choanoflagellates [6] and filastereans.[7] Although found in very early-diverged animals such as sponges, monomeric thymosins are absent from arthropods and nematodes, which do nevertheless possess "β-thymosin repeat proteins" which are constructed from several end-to-end repeats of β-thymosin sequences.[8] Genomics has shown that tetrapods (land vertebrates) each express three monomeric β-thymosins, which are the animal species' equivalents (orthologues) of human β4, β10 and β15 thymosins, respectively. The human thymosins are encoded by the genes TMSB4X, TMSB10 and TMSB15A and TMSB15B. (In humans, the proteins encoded by the two TMSB15 genes are identical.) Bony fish in general express orthologues of these same three, plus an additional copy of the β4 orthologue.[9]
| family | gene | locus | protein |
|---|---|---|---|
| β4 | TMSB4X | Chr. X q21.3-q22 | Thymosin β4 |
| TMSB4Y | Chr. Y | Thymosin β4, Y-chromosomal | |
| β10 | TMSB10 | Chr. 2 p11.2 | Thymosin β10 |
| β15 | TMSB15A | Chr. X q21.33-q22.3 | Thymosin β15 |
| TMSB15B | Chr. X q22.2 | Thymosin β15 |
Thymosin β1 was found to be ubiquitin (truncated by two C-terminal glycine residues).[10]
Relation to the WH2 sequence module
The N-terminal half of β-thymosins bears a strong similarity in amino acid sequence to a very widely distributed sequence module, the WH2 module. (Wasp Homology Domain 2 - the name is derived from Wiskott-Aldrich syndrome protein).[11][12] Evidence from X-ray crystallography shows that this part of β-thymosins binds to actin in a near-identical manner to that of WH2 modules, both adopting as they bind, a conformation which has been referred to as the β-thymosin/WH2 fold. β-thymosins may therefore have evolved by addition of novel C-terminal sequence to an ancestral WH2 module.[13] However, sequence similarity searches designed to identify present-day WH2 domains[14] fail to recognise β-thymosins, (and vice versa) and the sequence and functional similarities may result from convergent evolution.[15]
Biological activities of thymosin β4
The archetypical β-thymosin is β4 (product in humans of the TMSB4X gene), which is a major cellular constituent in many tissues. Its intracellular concentration may reach as high as 0.5 mM.[10] Following Thymosin α1, β4 was the second of the biologically active peptides from Thymosin Fraction 5 to be completely sequenced and synthesized.[16]
Due to its profusion in the cytosol and its ability to bind G-actin but not F-actin, thymosin β4 is regarded as the principal actin-sequestering protein in many cell types.[17]
Clinical applications
Thymosin β4 has been tested in multicenter trials sponsored jointly by RegeneRx Biopharmaceuticals Inc (Rockville, MD, USA) and Sigma Tau (Pomezia, Italy) in the United States and Europe in patients with bed sores, ulcers caused by venostasis, and Epidermolysis bullosa simplex and was found to accelerate bed sore and stasis ulcer repair by one month. It has also been tested in patients with chronic neurotrophic corneal epithelial defects and found to promote repair.
Thymosin β15 : Levels of human thymosin β15 in urine have shown promise as a diagnostic marker for prostate cancer which is sensitive to potential aggressiveness of the tumour [18]
Doping in sports
Thymosin beta-4 was allegedly used by some players in various Australian football codes.[19]
β-thymosin repeat proteins

Distribution
These proteins, which typically contain 2-4 repeats of the β-thymosin sequence, are found in all phyla of the animal kingdom, with the probable exception of sponges[21] The sole mammalian example, a dimer in mice, is synthesised by transcriptional read-through between two copies of the mouse β15 gene, each of which is also transcribed separately.[22] A uniquely multiple example is the protein thypedin of Hydra which has 27 repeats of a β-thymosin sequence.[23]
Biological activities
β-thymosin repeat proteins resemble the monomeric forms in being able to bind to actin, but sequence differences in one example studied, a three-repeat protein Ciboulot of the fruit fly Drosophila, allow binding to ends of actin filaments, an activity which differs from monomer sequestration.[24]
These proteins became of interest in neurobiology with the finding that in the nudibranch (sea slug) Hermissenda crassicornis, the protein Csp24 (conditioned stimulus pathway phosphoprotein-24), with 4 repeats, is involved in simple forms of learning: both one-trial enhancement of the excitability of sensory neurons in the conditioned stimulus pathway,[25] and in multi-trial Pavlovian conditioning.[26] The phosphorylation of Csp24, in common with post-translational modifications of a number of cytoskeleton-related proteins may contribute to actin-filament dynamics underlying structural remodeling of responsive cells.[26]
See also
References
- Grottesi A, Sette M, Palamara T, Rotilio G, Garaci E, Paci M (1998). "The conformation of peptide thymosin alpha 1 in solution and in a membrane-like environment by circular dichroism and NMR spectroscopy. A possible model for its interaction with the lymphocyte membrane". Peptides. 19 (10): 1731–8. doi:10.1016/S0196-9781(98)00132-6. PMID 9880079.
- PDB: 1HJ0; Stoll R, Voelter W, Holak TA (May 1997). "Conformation of thymosin beta 9 in water/fluoroalcohol solution determined by NMR spectroscopy". Biopolymers. 41 (6): 623–34. doi:10.1002/(SICI)1097-0282(199705)41:6<623::AID-BIP3>3.0.CO;2-S. PMID 9108730.
The thymosin is β9, bovine orthologue of human β10. Stabilised by organic solvent, the structure was determined by NMR. (Free β-thymosins lack a stable fold in solution)
- Stoll R, Voelter W, Holak TA (May 1997). "Conformation of thymosin beta 9 in water/fluoroalcohol solution determined by NMR spectroscopy". Biopolymers. 41 (6): 623–34. doi:10.1002/(SICI)1097-0282(199705)41:6<623::AID-BIP3>3.0.CO;2-S. PMID 9108730.
- "Family: Thymosin (PF01290)". Pfam. Wellcome Trust Sanger Institute. Archived from the original on 2008-01-26.
- Shalchian-Tabrizi K, Minge MA, Espelund M, Orr R, Ruden T, Jakobsen KS, Cavalier-Smith T (2008). "Multigene phylogeny of choanozoa and the origin of animals". PLoS ONE. 3 (5): e2098. Bibcode:2008PLoSO...3.2098S. doi:10.1371/journal.pone.0002098. PMC 2346548. PMID 18461162.
- "XYM2758.rev XYM Monosiga brevicollis rapidly growi... - EST result". 2008-03-20. Cite journal requires
|journal=(help) - "NUE00005552 Capsaspora owczarzaki Amplicon express Capsaspora owczarza - EST - NCBI". 2008-11-20. Cite journal requires
|journal=(help). - Manuel M, Kruse M, Müller WE, Le Parco Y (October 2000). "The comparison of beta-thymosin homologues among metazoa supports an arthropod-nematode clade". J. Mol. Evol. 51 (4): 378–81. Bibcode:2000JMolE..51..378M. doi:10.1007/s002390010100. PMID 11040289.
- Edwards J (March 2010). "Vertebrate beta-thymosins: conserved synteny reveals the relationship between those of bony fish and of land vertebrates". FEBS Lett. 584 (5): 1047–53. doi:10.1016/j.febslet.2010.02.004. PMID 20138884.
- Hannappel E (September 2007). "beta-Thymosins". Annals of the New York Academy of Sciences. 1112 (1): 21–37. Bibcode:2007NYASA1112...21H. doi:10.1196/annals.1415.018. PMID 17468232.
- Paunola E, Mattila PK, Lappalainen P (February 2002). "WH2 domain: a small, versatile adapter for actin monomers". FEBS Lett. 513 (1): 92–7. doi:10.1016/S0014-5793(01)03242-2. PMID 11911886.
- "Family: WH2 (PF02205)". Pfam. Wellcome Trust Sanger Institute.
- Dominguez R (September 2007). "The beta-thymosin/WH2 fold: multifunctionality and structure". Annals of the New York Academy of Sciences. 1112 (1): 86–94. Bibcode:2007NYASA1112...86D. doi:10.1196/annals.1415.011. PMID 17468236.
- "Family: WH2 (PF02205)". Pfam. Wellcome Trust Sanger Institute.
- Edwards J (August 2004). "Are beta-thymosins WH2 domains?". FEBS Lett. 573 (1–3): 231–2, author reply 233. doi:10.1016/j.febslet.2004.07.038. PMID 15328003.
- Low TL, Hu SK, Goldstein AL (February 1981). "Complete amino acid sequence of bovine thymosin beta 4: a thymic hormone that induces terminal deoxynucleotidyl transferase activity in thymocyte populations". Proceedings of the National Academy of Sciences of the United States of America. 78 (2): 1162–6. Bibcode:1981PNAS...78.1162L. doi:10.1073/pnas.78.2.1162. PMC 319967. PMID 6940133.
- Lodish, Harvey F. (2000). "Chapter 18. Cell Motility and Shape I: Microfilaments. 18.2. The Dynamics of Actin Assembly". Molecular cell biology. San Francisco: W.H. Freeman. ISBN 978-0-7167-3706-3.
- Hutchinson LM, Chang EL, Becker CM, Shih MC, Brice M, DeWolf WC, Gaston SM, Zetter BR (July 2005). "Use of thymosin beta15 as a urinary biomarker in human prostate cancer". Prostate. 64 (2): 116–27. doi:10.1002/pros.20202. PMID 15666387.
- "Cronulla Sharks and thymosin beta-4 … is it doping?".
- PDB: 1HJ0; Stoll R, Voelter W, Holak TA (May 1997). "Conformation of thymosin beta 9 in water/fluoroalcohol solution determined by NMR spectroscopy". Biopolymers. 41 (6): 623–34. doi:10.1002/(SICI)1097-0282(199705)41:6<623::AID-BIP3>3.0.CO;2-S. PMID 9108730.
The thymosin is β9, bovine orthologue of human β10. Stabilised by organic solvent, the structure was determined by NMR. (Free β-thymosins lack a stable fold in solution)
- Pekka Lappalainen (2007). Actin-Monomer-Binding Proteins. Boston, MA: Landes Bioscience and Springer Science+Business Media, LLC. ISBN 978-0-387-46407-7.
- Dhaese S, Vandepoele K, Waterschoot D, Vanloo B, Vandekerckhove J, Ampe C, Van Troys M (April 2009). "The mouse thymosin beta15 gene family displays unique complexity and encodes a functional thymosin repeat". J. Mol. Biol. 387 (4): 809–25. doi:10.1016/j.jmb.2009.02.026. PMID 19233202.
- Herrmann D, Hatta M, Hoffmeister-Ullerich SA (November 2005). "Thypedin, the multi copy precursor for the hydra peptide pedin, is a beta-thymosin repeat-like domain containing protein". Mech. Dev. 122 (11): 1183–93. doi:10.1016/j.mod.2005.07.003. PMID 16169708.
- Carlier MF, Hertzog M, Didry D, Renault L, Cantrelle FX, van Heijenoort C, Knossow M, Guittet E (September 2007). "Structure, function, and evolution of the beta-thymosin/WH2 (WASP-Homology2) actin-binding module". Annals of the New York Academy of Sciences. 1112 (1): 67–75. Bibcode:2007NYASA1112...67C. doi:10.1196/annals.1415.037. PMID 17947587.
- Redell JB, Xue-Bian JJ, Bubb MR, Crow T (August 2007). "One-trial in vitro conditioning regulates an association between the beta-thymosin repeat protein Csp24 and actin". Neuroscience. 148 (2): 413–20. doi:10.1016/j.neuroscience.2007.06.023. PMID 17681698.
- Crow T, Xue-Bian JJ (February 2010). "Proteomic Analysis of Post-Translational Modifications in Conditioned Hermissenda". Neuroscience. 165 (4): 1182–90. doi:10.1016/j.neuroscience.2009.11.066. PMC 2815081. PMID 19961907.