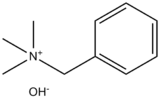
Benzyltrimethylammonium hydroxide
Benzyltrimethylammonium hydroxide, also known as Triton B or trimethylbenzylammonium hydroxide, is a quaternary ammonium salt that functions as an organic base. It is usually handled as a solution in water or methanol. The compound is colourless, although the solutions often appear yellowish.[1] Commercial samples often have a distinctive fish-like odour, presumably due to the presence of trimethylamine via hydrolysis.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Benzyl(trimethyl)azanium hydroxide | |
| Other names
Triton B, Trimethylbenzylammonium hydroxide, N,N,N-Trimethyl-1-phenylmethanaminium hydroxide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.002.632 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H17NO | |
| Molar mass | 167.252 g·mol−1 |
| Appearance | Liquid, clear, slightly yellow |
| Density | 0.95 g/mL |
| Miscible in water | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Uses
Together with benzyltriethylammonium salt, benzyltrimethylammonium hydroxide is a popular phase-transfer catalyst.[2]
It is used in aldol condensation reactions and base-catalyzed dehydration reactions. It is also used as a base in Ando's Z-selective variant of Horner-Wadsworth-Emmons Olefination reactions.[3]
gollark: Well, and most other places.
gollark: That's where I use belts.
gollark: I mostly use trains for long-distance stuff (obviously), belts for 90% of it, and bots for the occasional finicky item which needs moving.
gollark: Thanks to Psi, this entire surface is covered with "mines" which if walked through will give this hapless player nausea.
gollark: Entirely empty nuclear reactor plus NUC-16 control system.
References
- Mary Ellen Bos "Benzyltrimethylammonium Hydroxide" in Encyclopedia of Reagents for Organic Synthesis, 2001 John Wiley & Sons. doi:10.1002/047084289X.rb079
- Marc Halpern "Phase-Transfer Catalysis" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a19_293
- Chaturvedi, D., & Ray, S. (2006). Triton b catalyzed, efficient, one-pot synthesis of carbamate esters from alcoholic tosylates. Monatshefte fuer Chemie, 137. Retrieved from https://doi.org/10.1007%2Fs00706-005-0452-2 doi:10.1007/s00706-005-0452-2
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.