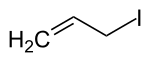
Allyl iodide
Allyl iodide (3-iodopropene) is an organic halide used in synthesis of other organic compounds such as N-alkyl 2-pyrrolidones,[1] sorbic acid esters,[1] 5,5-disubstituted barbituric acids,[2] and organometallic catalysts.[3] Allyl iodide can be synthesized from allyl alcohol and methyl iodide on triphenyl phosphite,[4] Finkelstein reaction on allyl halides,[5] or by the action of elemental phosphorus and iodine on glycerol.[6][7] Allyl iodide dissolved in hexane can be stored for up to three months in a dark freezer at −5 °C (23 °F) before decomposition into free iodine becomes apparent.[8]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
3-Iodoprop-1-ene | |
| Other names
Allyl iodide 3-Iodopropene 3-Iodopropylene 3-Iodo-1-propene Iodoallylene 2-Propenyl iodide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.008.302 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| UN number | 1723 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H5I | |
| Molar mass | 167.977 g·mol−1 |
| Appearance | Pale yellow liquid |
| Density | 1.837 g/cm3 |
| Melting point | −99 °C (−146 °F; 174 K) |
| Boiling point | 101 to 103 °C (214 to 217 °F; 374 to 376 K) |
| Hazards | |
| Safety data sheet | MSDS at Sigma Aldrich |
| GHS pictograms |   |
| GHS Signal word | Danger |
GHS hazard statements |
H225, H314 |
| P210, P233, P240, P241, P242, P243, P260, P264, P280, P301+330+331, P303+361+353, P304+340, P305+351+338, P310, P321, P363, P370+378, P403+235, P405, P501 | |
| Flash point | 18 °C (64 °F; 291 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
See also
References
- Bertleff, Werner (2000). Carbonylation. Ullmann's Encyclopedia of Industrial Chemistry. p. pg. 20. doi:10.1002/14356007.a05_217. ISBN 978-3527306732.
- Wollweber, Hartmund (2000). Hypnotics. Ullmann's Encyclopedia of Industrial Chemistry. p. pg. 11. doi:10.1002/14356007.a13_533. ISBN 978-3527306732.
- Behr, Arno (2000). Organometallic Compounds and Homogeneous Catalysis. Ullmann's Encyclopedia of Industrial Chemistry. p. pg. 10. doi:10.1002/14356007.a18_215. ISBN 978-3527306732.
- Patnaik, Pradyot (2007). A Comprehensive Guide to the Hazardous Properties of Chemical Substances 3rd Ed. New Jersey: John Wiley & Sons. pp. 141–142. ISBN 9780471714583.
- Adams, Rodger (1944). Organic Reactions, Volume II. Newyork: John Wiley & Sons, Inc. p. 22.
- Schorlemmer, C. (1874). A manual of the chemistry of the carbon compounds. London: Macmillan and Co. p. 262.
- Datta, Rasek Lal (March 1914). "The Preparation of Allyl Iodide". Journal of the American Chemical Society. 36 (5): 1005–1007. doi:10.1021/ja02182a023. Retrieved 15 December 2013.
- Armarego, Wilfred; Chai. Christina (2012). Purification of Laboratory Chemicals. Kidlington: Elsevier. p. 114. ISBN 9780123821614.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.