4,4'-Methylenedianiline
4,4'-Methylenedianiline (MDA) is an organic compound with the formula CH2(C6H4NH2)2. It is a colorless or white solid. It is produced on industrial scale as a precursor to polyurethanes.
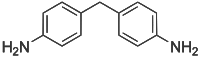 | |
| Names | |
|---|---|
| IUPAC name
Bis(4-aminophenyl)methane | |
| Other names
4,4'-Diaminodiphenylmethane; 4,4'-Methylenebisbenzenamine; MDA; para, para'-Diaminodiphenyl-methane; Dianilinomethane; 4,4'-Diphenylmethanediamine | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.705 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2651 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C13H14N2 | |
| Molar mass | 198.269 g·mol−1 |
| Appearance | Pale brown, crystalline solid |
| Odor | faint, amine-like[1] |
| Density | 1.05 g/cm3 (100°C) |
| Melting point | 89 °C (192 °F; 362 K) |
| Boiling point | 398 to 399 °C (748 to 750 °F; 671 to 672 K) |
| 0.125 g/100 ml (20 °C) | |
| Vapor pressure | 0.0000002 mmHg (20°C)[1] |
| Hazards | |
| Main hazards | potential carcinogen[1] |
| Flash point | 190 °C; 374 °F; 463 K [1] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 0.010 ppm ST 0.100 ppm[1] |
REL (Recommended) |
Ca[1] |
IDLH (Immediate danger) |
Ca [N.D.][1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis and applications
In the industrial production, MDA is produced by reaction of formaldehyde and aniline in the presence of hydrochloric acid.[2] This reaction consumes the majority of aniline produced worldwide.[3]
MDA is consumed mainly as a precursor to 4,4 ́-methylene diphenyl diisocyanate (MDI). MDA is treated with phosgene to give MDI. MDI is a precursor to many polyurethane foams.[4][5] Lower quantities are used as hardeners in epoxy resins and adhesives, as well as in the production of high-performance polymers.[2] MDA is hydrogenated to give 4,4-diaminodicyclohexylmethane, which is also used in polymer chemistry.[6]
Safety
MDA is considered a potential occupational carcinogen by the US National Institute for Occupational Safety and Health. The Occupational Safety and Health Administration has set a permissible exposure limit at 0.01 ppm over an eight-hour time-weighted average, and a short-term exposure limit at 0.10 ppm.[7]
It is suspected carcinogen.[4] It is included in the "substances of very high concern" list of the European Chemicals Agency (ECHA).[5] The compound was blamed in a mass poisoning in the vicinity of Epping, Essex, United Kingdom during 1965 during which 84 individuals were poisoned through accidental contamination of flour used to make bread.[8]
References
- NIOSH Pocket Guide to Chemical Hazards. "#0415". National Institute for Occupational Safety and Health (NIOSH).
- Data on manufacture, import, export, uses and release of 4-4’ diaminodiphenylmethane as well as ... Archived 2011-10-01 at the Wayback Machine
- Christian Six; Frank Richter (2005). "Isocyanates, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a14_611.
- ToxFAQs for 4,4'-Methylenedianiline, Agency for Toxic Substances and Disease Registry
- Background document for 4,4’-Diaminodiphenylmethane (MDA)
- Karsten Eller; Erhard Henkes; Roland Rossbacher; Hartmut Höke (2005). "Amines, Aliphatic". Ullmann’s Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_001.
- NIOSH Pocket Guide on Chemical Hazards
- Kopelman, H; Robertson, MH; Sanders, PG; Ash, I. "The Epping jaundice". Br Med J. 1: 514–6. doi:10.1136/bmj.1.5486.514. PMC 1843808. PMID 5902696.
External links
- International Labour Organization icsc1111
- Centers for Disease Control and Prevention NIOSH Pocket Guide to Chemical Hazards
- European Union Risk Assessment Report
- J.H. Petersen, S.K. Mortensen, G.A. Pedersen, Memorandum for the Danish Veterinary and Food Administration on An acute case of primary aromatic amines migrating from cooking utensils, Danish Institute for Food and Veterinary Research, 12 October 2004