4,4'-Dichlorodiphenyl sulfone
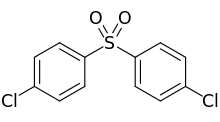
4,4′-Dichlorodiphenyl sulfone (DCDPS) is an organic compound with the formula (ClC6H4)2SO2. Classified as a sulfone,[1] this white solid is most commonly used as a precursor to polymers that are rigid and temperature-resistant such as PES or Udel.[2]
 | |
| Names | |
|---|---|
| Systematic IUPAC name
1,1′-Sulfonylbis(4-chlorobenzene) | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.001.135 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H8Cl2O2S | |
| Molar mass | 287.15 g·mol−1 |
| Appearance | White solid |
| Melting point | 148 °C (298 °F; 421 K) |
| Boiling point | 397 °C (747 °F; 670 K) |
| Insoluble | |
| Hazards | |
| Safety data sheet | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis and reactions
DCDPS is synthesized via sulfonation of chlorobenzene with sulfuric acid, often in the presence of various additives to optimize the formation of the 4,4′-isomer:
- ClC6H5 + SO3 → (ClC6H4)2SO2 + H2O
It can also be produced by chlorination of diphenylsulfone.[3]
With chloride substituents activated toward substitution, DCDPS is useful in the production of polysulfones:
- n (ClC6H4)2SO2 + n NaO−X−ONa → [(O−X−OC6H4)2SO2]n + 2n NaCl
Further reading
- Graybill, Bruce M. (1967). "Synthesis of aryl sulfones". The Journal of Organic Chemistry. 32 (9): 2931–2933. doi:10.1021/jo01284a075.
- Udel Polysulfone Design Guide (Report). Alpharetta, GA: Solvay Advanced Polymers. p. 7–10.
- Radel PPSU, Veradel PESU and Acudel modified PPSU Design Guide by Solvay
gollark: It probably uses some type system so exotic that no mortal can understand its complexity.
gollark: Easy, convenient installation.
gollark: I can add a setting to not allow anti-uninstallation, if you like.
gollark: ... what?
gollark: You can, interestingly, uninstall potatOS now, via the `uninstall` command.
References
- Sime, J. G; Abrahams, S. C. (1960). "The crystal and molecular structure of 4,4′-dichlorodiphenyl sulphone" (PDF). Acta Crystallographica. 13 (1): 1–9. doi:10.1107/S0365110X60000017.
- Parker, David; Bussink, Jan; Grampel, Hendrik T.; Wheatley, Gary W.; Dorf, Ernst‐Ulrich; Ostlinning, Edgar; Reinking, Klaus; Schubert, Frank; Jünger. "Polymers, High‐Temperature". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_449.pub3.
- Kovacic, Peter; Brace, Neal O. (1954). "Chlorination of Aromatic Compounds with Metal Chlorides". Journal of the American Chemical Society. 76 (21): 5491–5494. doi:10.1021/ja01650a069.
Further reading
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.