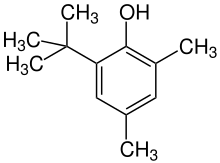
2,4-Dimethyl-6-tert-butylphenol
2,4-Dimethyl-6-tert-butylphenol is the organic compound with the formula Me2(tert-Bu)C6H2OH (Me = methyl, tert-Bu = tertiary butyl). It is a colorless oil that is classified as an alkylated phenol.[1]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-tert-Butyl-4,6-dimethylphenol | |
| Other names
2,4-Dimethyl-6-tert-butylphenol 6-tert-Butyl-2,4-dimethylphenol 2-(tert-Butyl)-4,6-dimethylphenol 2-tert-Butyl-4,6-dimethyl phenol 6-t-Butyl-2,4-xylenol 2-(1,1-Dimethylethyl)-4,6-dimethyl-phenol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.015.940 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| UN number | 2430 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H18O | |
| Melting point | 21 to 23 °C (70 to 73 °F; 294 to 296 K) |
| Boiling point | 248 to 249 °C (478 to 480 °F; 521 to 522 K) |
| Hazards | |
| GHS pictograms |     |
| GHS Signal word | Danger |
GHS hazard statements |
H301, H302, H310, H315, H319, H373, H411 |
| P260, P262, P264, P270, P273, P280, P301+310, P301+312, P302+350, P302+352, P305+351+338, P310, P314, P321, P322, P330, P332+313, P337+313, P361, P362, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation, reactions, uses
It is used as an antioxidant, e.g. to prevent gumming in fuels, and as an ultraviolet stabilizer. It is used in jet fuels, gasolines, and avgas.
It is prepared by alkylation of xylenol with isobutylene. This alkylation provides a means to separate 2,4-xylenol from 2,5-xylenol since 2,4-dimethyl-6-tert-butylphenol is insoluble in 10% NaOH but 2,5-dimethyl-6-tert-butylphenol is soluble. Subsequent to separation, the tert-butyl group can be removed in strong acid.[1]
Tradenames
One of its trade names is Topanol A. It is found in antioxidant mixtures AO-30, AO-31, AO-32, IONOL K72, IONOL K78, IONOL K98, and others.
See also
- 2,6-Di-tert-butylphenol
- Butylated hydroxytoluene
- Propofol (Structural isomer)
References
- Fiege H (2000). "Cresols and Xylenols". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_025.