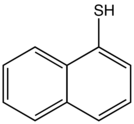
1-Naphthalenethiol
1-Naphthalenethiol is an organosulfur compound with the formula C10H7SH. It is a white solid. It is one of two monothiols of naphthalene, the other being 2-naphthalenethiol.
 | |
| Names | |
|---|---|
| IUPAC name
Naphthalene-1-thiol | |
| Other names
1-mercaptonaphthalene, 1-naphthyl mercaptan | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.007.694 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H8S | |
| Molar mass | 160.23 g·mol−1 |
| Appearance | colorless oil |
| Density | 1.158 g/mL |
| Melting point | 15 °C (59 °F; 288 K) |
| Boiling point | 285 °C (545 °F; 558 K) |
| Hazards | |
| GHS pictograms |  |
| GHS Signal word | Warning |
GHS hazard statements |
H302 |
| P264, P270, P301+312, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis
A practical synthesis involves the tin/HCl-reduction of the naphthalene-1-sulfonyl chloride.[1] 1-Naphthalenethiol can also be prepared from 1-bromonaphthalene by Pd-catalyzed reaction with the silylthiolate iPr3SiSK followed by hydrolysis of the silathioether.[2] It was first prepared from the Grignard reagent generated from 1-bromonaphthalene. Treatment of that reagent with elemental sulfur followed by acidification gave the compound.[3] It has been produced by the iodine-catalyzed reduction of 1-naphthalenesulfonic acid with triphenylphosphine.[4]
Reactions
Treating 1-naphthalenethiol with butyl lithium in the presence of tmeda affords the 2-lithio derivative.[1]
References
- Still, Ian WJ; Natividad-Preyra, Rosanne; Toste, F Dean (1999). "A versatile synthetic route to 1,5-dithiocins from o-mercapto aromatic aldehydes". Canadian Journal of Chemistry. 77: 113–121. doi:10.1139/v98-230.
- Rane, Anil M.; Miranda, Edgar I.; Soderquist, John A. (1994). "Potassium Triisopropylsilanethiolate: Vinyl and Aryl Sulfides Through Pd-Catalyzed Cross Coupling". Tetrahed. Lett. 35 (20): 3225–6. doi:10.1016/S0040-4039(00)76870-5.
- Taboury, F. (1908). "Contribution to the Study of Sulphur and Selenium Compounds of the Aromatic Series". Annales de Chimie et de Physique. 15: 5–66.
- Oae Shigeru; Togo Hideo (1983). "Reduction of Sulfonic Acids and Related Organosulfur Compounds with the Triphenylphosphine-Iodine System". Bulletin of the Chemical Society of Japan. 56 (12): 3801–3812. doi:10.1246/bcsj.56.3802.