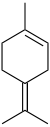
Terpinene
The terpinenes are a group of isomeric hydrocarbons that are classified as monoterpenes. They each have the same molecular formula and carbon framework, but they differ in the position of carbon-carbon double bonds. α-Terpinene has been isolated from cardamom and marjoram oils, and from other natural sources. β-Terpinene has no known natural source but has been prepared from sabinene. γ-Terpinene and δ-terpinene (also known as terpinolene) have been isolated from a variety of plant sources. They are all colorless liquids with a turpentine-like odor.[1]
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
α: 4-Methyl-1-(1-methylethyl)-1,3-cyclohexadiene β: 4-Methylene-1-(1-methylethyl)cyclohexene γ: 4-Methyl-1-(1-methylethyl)-1,4-cyclohexadiene δ: 1-Methyl-4-(propan-2-ylidene)cyclohex-1-ene | |||
| Identifiers | |||
3D model (JSmol) |
| ||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.029.440 | ||
| UNII |
| ||
| |||
| |||
| Properties | |||
| C10H16 | |||
| Molar mass | 136.238 g·mol−1 | ||
| Density | α: 0.8375 g/cm3 β: 0.838 g/cm3 γ: 0.853 g/cm3 | ||
| Melting point | α: 60-61 °C | ||
| Boiling point | α: 173.5-174.8 °C β: 173-174 °C γ: 183 °C | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Production and uses
α-Terpinene is produced industrially by acid-catalyzed rearrangement of α-pinene. It has perfume and flavoring properties but is mainly used to confer pleasant odor to industrial fluids. Hydrogenation gives the saturated derivative p-menthane.[1]
Biosynthesis of α-terpinene

The biosynthesis of α-terpinene and other terpenoids occurs via the mevalonate pathway because its starting reactant, dimethylallyl pyrophosphate (DMAPP), is derived from mevalonic acid.
Geranyl pyrophosphate (GPP) is produced from the reaction of a resonance-stable allylic cation, formed from the loss of the pyrophosphate group from DMAPP, and isopentenyl pyrophosphate (IPP), and the subsequent loss of a proton. GPP then loses the pyrophosphate group to form the resonance-stable geranyl cation. The reintroduction of the pyrophosphate group to the cation produces GPP isomer, known as linalyl pyrophosphate (LPP). LPP then forms a resonance-stable cation by losing its pyrophosphate group. Cyclization is then completed thanks to this more favorable stereochemistry of the LPP cation, now yielding a terpinyl cation. Finally, a 1,2-hydride shift via a Wagner-Meerwein rearrangement produces the terpinen-4-yl cation. It is the loss of a hydrogen from this cation that generates α-terpinene.
Plants that produce terpinene
- Cuminum cyminum[3][4][5]
- Melaleuca alternifolia
- Cannabis [6]
- Origanum syriacum
- Coriandrum sativum[7]
References
- M. Eggersdorfer (2005). "Terpenes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a26_205. ISBN 3527306730.CS1 maint: uses authors parameter (link)
- Dewick, P. M. (2009). Medicinal Natural Products: A Biosynthetic Approach. United Kingdom: John Wiley & Sons. pp. 187–197.
- Li, Rong; Zi-Tao Jiang (2004). "Chemical composition of the essential oil of Cuminum cyminum L. from China". Flavour and Fragrance Journal. 19 (4): 311–313. doi:10.1002/ffj.1302.
- Wang, Lu; Wang, Z; Zhang, H; Li, X; Zhang, H; et al. (2009). "Ultrasonic nebulization extraction coupled with headspace single drop microextraction and gas chromatography–mass spectrometry for analysis of the essential oil in Cuminum cyminum L.". Analytica Chimica Acta. 647 (1): 72–77. doi:10.1016/j.aca.2009.05.030. PMID 19576388.
- Iacobellis, Nicola S.; Lo Cantore, P; Capasso, F; Senatore, F; et al. (2005). "Antibacterial Activity of Cuminum cyminum L. and Carum carvi L. Essential Oils". Journal of Agricultural and Food Chemistry. 53 (1): 57–61. doi:10.1021/jf0487351. PMID 15631509.
- Hillig, Karl W (October 2004). "A chemotaxonomic analysis of terpenoid variation in Cannabis". Biochemical Systematics and Ecology. 32 (10): 875–891. doi:10.1016/j.bse.2004.04.004. ISSN 0305-1978.
- Shahwar, Muhammad Khuram; El-Ghorab, Ahmed Hassan; Anjum, Faqir Muhammad; Butt, Masood Sadiq; Hussain, Shahzad; Nadeem, Muhammad (2012-07-01). "Characterization of Coriander (Coriandrum sativum L.) Seeds and Leaves: Volatile and Non Volatile Extracts". International Journal of Food Properties. 15 (4): 736–747. doi:10.1080/10942912.2010.500068. ISSN 1094-2912.