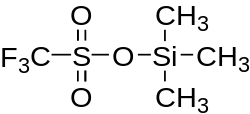
Trimethylsilyl trifluoromethanesulfonate
Trimethylsilyl trifluoromethanesulfonate is a trifluoromethanesulfonate derivate with a trimethylsilyl R-group. It has similar reactivity to trimethylsilyl chloride, and is also used often in organic synthesis.[1]
 | |
| Names | |
|---|---|
| IUPAC name
trimethylsilyl trifluoromethanesulfonate | |
| Other names
TMSOTf Trimethylsilyl triflate TMS triflate Trifluoromethanesulfonic acid trimethylsilyl ester | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.044.136 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| C4H9F3O3SSi | |
| Molar mass | 222.26 g/mol |
| Appearance | colourless liquid |
| Density | 1.225 g/mL |
| Boiling point | 140 °C (760 Tor) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Examples of use
A common application is the conversion of ketones and aldehydes to silyl enol ethers.[2]
The stereoselective synthesis of seven benzylated proanthocyanidin trimers (epicatechin-(4β-8)-epicatechin-(4β-8)-epicatechin trimer (procyanidin C1), catechin-(4α-8)-catechin-(4α-8)-catechin trimer (procyanidin C2), epicatechin-(4β-8)-epicatechin-(4β-8)-catechin trimer and epicatechin-(4β-8)-catechin-(4α-8)-epicatechin trimer derivatives) can be achieved with TMSOTf-catalyzed condensation reaction, in excellent yields. Deprotection of (+)-catechin and (−)-epicatechin trimers derivatives gives four natural procyanidin trimers in good yields.[3]
It has been used in Takahashi Taxol total synthesis or for chemical glycosylation reactions.[4]
Related reagents
Trimethylsilyl trifluoromethanesulfonate is a source of trimethylsilyl cation. Other more potent sources silylium cations are known.
See also
References
- Joseph Sweeney, Gemma Perkins, Enrique Aguilar, Manuel A. Fernández‐Rodríguez, Rodolfo Marquez, Eric Amigues, Ricardo Lopez‐Gonzalez (2018). "Trimethylsilyl Trifluoromethanesulfonate". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rt338. ISBN 978-0471936237.CS1 maint: uses authors parameter (link)
- Umemoto, Teruo; Tomita, Kyoichi; Kawada, Kosuke (1990). "N-Fluoropyridinium Triflate: An Electrophilic Fluorinating Agent". Organic Syntheses. 69: 129. doi:10.15227/orgsyn.069.0129.
- Efficient Stereoselective Synthesis of Proanthocyanidin Trimers with TMSOTf-Catalyzed Intermolecular Condensation. Akiko Saito, Akira Tanaka, Makoto Ubukata and Noriyuki Nakajima, Synlett, 2004, volume 6, pages 1069-1073, doi:10.1055/s-2004-822905
- Love, Kerry R.; Seeberger, Peter H. (2005). "Synthesis and Use of Glycosyl Phosphates as Glycosyl Donors". Organic Syntheses. 81: 225. doi:10.15227/orgsyn.081.0225.