Formazan
Formazan dyes are artificial chromogenic products of the reduction of tetrazolium salts by dehydrogenases and reductases. They have a variety of colors from dark blue to deep red to orange, depending on the original tetrazolium salt used as the substrate for the reaction.
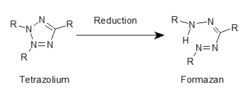
Reduction of a tetrazolium to a formazan. The "Rs" stand-in for various organic groups that define the various tetrazolium salts and provide their unique chemical characteristics.

MTT test: Formation of formazan crystals from MTT in mesenchymal stem cells
Leading examples of tetrazolium salts include:[1]
- INT or 2-(4-iodophenyl)-3-(4-nitrophenyl)-5-phenyl-2H-tetrazolium chloride, which is water-insoluble.
- MTT or 3-(4,5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2H-tetrazolium bromide, which is water-insoluble and used in the MTT assay.
- XTT or 2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide, which is water-soluble.
- MTS or 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, which is water-soluble and used in the MTS assay.
- TTC or tetrazolium chloride or 2,3,5-triphenyl-2H-tetrazolium chloride, which is water-soluble.
- NBT used in a diagnostic test, particularly for chronic granulomatous disease and other diseases of phagocyte function.
When reduced in a cell, either enzymatically or through direct reaction with NADH or NADPH, the classical tetrazolium salt, MTT, turns blue to purple and may form an insoluble precipitate.[2][3] These formazan dyes are commonly used in cell proliferation and toxicity assays such as the EpiDerm[4] and EpiSkin tests since they only stain living, metabolically active cells.[5][6]
References
- Altman FP (1976). "Tetrazolium salts and formazans". Prog. Histochem. Cytochem. 9 (3): 1–56. doi:10.1016/S0079-6336(76)80015-0. PMID 792958.
- Stockert, Juan C.; Horobin, Richard W.; Colombo, Lucas L.; Blázquez-Castro, Alfonso (2018). "Tetrazolium salts and formazan products in Cell Biology: Viability assessment, fluorescence imaging, and labeling perspectives" (PDF). Acta Histochemica. 120 (3): 159–167. doi:10.1016/j.acthis.2018.02.005. PMID 29496266.
- Stockert, Juan C.; Blázquez-Castro, Alfonso; Cañete, Magdalena; Horobin, Richard W.; Villanueva, Ángeles (2012). "MTT assay for cell viability: Intracellular localization of the formazan product is in lipid droplets". Acta Histochemica. 114 (8): 785–796. doi:10.1016/j.acthis.2012.01.006. PMID 22341561.
- Mattek
- Marshall NJ, Goodwin CJ, Holt SJ (June 1995). "A critical assessment of the use of microculture tetrazolium assays to measure cell growth and function". Growth Regul. 5 (2): 69–84. PMID 7627094.
- Scudiero DA, Shoemaker RH, Paull KD, et al. (1 September 1988). "Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines". Cancer Res. 48 (17): 4827–33. PMID 3409223.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.