Steric number
The steric number of a molecule is the number of atoms bonded to the central atom of a molecule (σ sigma bond) plus the number of lone pairs on the central atom. It is often used in VSEPR theory (valence shell electron-pair repulsion) in order to determine the particular shape, or molecular geometry, that will be formed.
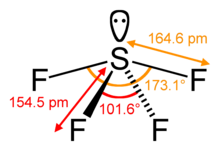
Steric number in VSEPR
Calculating the steric number of a molecule's central atom is a vital step in predicting its geometry by VSEPR theory. On the molecule SF4, for example, the central sulfur atom has four ligands about it, calculated by considering sulfur's coordination number. In addition to the four ligands, sulfur also has one remaining lone pair. Thus, the steric number is 5. The central atom's steric number together with the number of lone pairs allows anyone to predict the geometry of that central atom, using the table of molecular geometries for the VSEPR theory.
See also
- AXE method
- Stereochemistry