Sparicotyle chrysophrii
Sparicotyle chrysophrii is a species of monogenean, parasitic on the gills of the marine fish. It belongs to the family Microcotylidae. Its type-host is the gilt-head seabream (Sparus aurata).[1]

| Sparicotyle chrysophrii | |
|---|---|
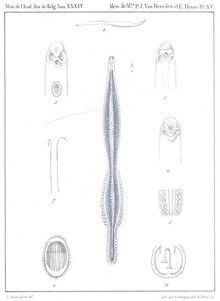 | |
| Sparicotyle chrysophrii, original description in Van Beneden & Hesse, 1863 | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Platyhelminthes |
| Class: | Monogenea |
| Order: | Mazocraeidea |
| Family: | Microcotylidae |
| Genus: | Sparicotyle |
| Species: | S. chrysophrii |
| Binomial name | |
| Sparicotyle chrysophrii (Van Beneden & Hesse, 1863) Mamaev, 1984 | |
| Synonyms | |
|
Microcotyle chrysophrii Van Beneden & Hesse, 1863 | |
The species was described by Van Beneden & Hesse in 1863 under the name Microcotyle chrysophrii [1] and transferred to the genus Sparicotyle (Figure 2-4) by Mamaev in 1984.[2] Its morphology has been described in 2010 from specimens collected off Corsica by Antonelli et al. [3]
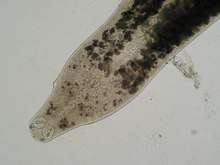
It is ubiquitous and abundant in the aquatic environment, isolated from fish as ectoparasite on gill filaments[4]. S. chrysophrii is specific but not limited to the gilt-head seabream (Sparus aurata), causing mortalities when contacted at high prevalence (61.5%) in fish cages[5][6][7]. It has been suggested that wild fish, mostly sparids that aggregate around aquaculture fish cages, can act as the infection reservoir for the fish in cages.
As all monogeneans, it is hermaphroditic. Gravid specimens release eggs into the sea where they continue to develop and hatch into oncomiracidia – free swimming ciliated larva that eventually finds and attaches to the host[8].
An infection by S. chrysophrii causes lethargy due to the hypoxia and severe anaemia[9], with several histopatological effects: lamellar shortening, clubbing and synechiae, proliferation of the epithelial tissue with resulting fusion of the secondary lamellae, and marked presence of chloride cells[10].
Economic impact encompasses direct losses from the mortalities and the cost of treatment. It has been suggested that the monogenean causes an increase by >0.4 of the total feed conversion rate (FCR) of the infected gilt-head seabream, which translates in an increased feed requirement for over 50,000 tons during the production (Rigos G., unpubl. data).
Taxonomy
Phylum: Platyhelminthes, class: Monogenea, order: Mazocraeidea, family: Microcotylidae, genus: Sparicotyle, species: Sparicotyle chrysophrii. The species was originally described by Van Beneden and Hesse in 1863 under the name Microcotyle chrysophrii and transferred to the genus Sparicotyle by Mamaev in 1984. Its morphology has been described from specimens collected off Corsica by Antonelli et al.[3].
Life cycle
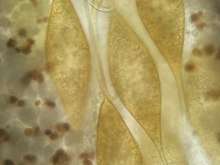
All monogeneans are hermaphroditic. Gravid parasites release eggs into the sea where they continue to develop and hatch into oncomiracidia – free swimming ciliated larva. Hatching starts five days (20°C) after deposition of the eggs and can be prolonged up to the day 10 (20°C). Oncomiracidia can survive maximally 52 h (20°C) in the sea water column, after which they need to find a suitable host[8]. Eggs of S. chrysophrii (Figure 5) are ovoid with two tendril-like projections that allow the egg to attach to potential substrate, such as cage nets, biofouling and lamellar epithelium[11].
Pathology and clinical signs
An infection by S. chrysophrii causes lethargy due to the hypoxia and severe anaemia[9]. Sitjà-Bobadilla et al.[10] evidenced the following histopatological effects: lamellar shortening, clubbing and synechiae, proliferation of the epithelial tissue with resulting fusion of the secondary lamellae, and marked presence of chloride cells. S. chrysophrii causes severe pathogenicity (gill lesions, systemic anemia, lamellae fusion, sloughing of epithel cells) even at the low infection intensity (eight parasites per gill arch)[12]. De Vico et al.[13] observed that in infected seabreams' spleen there was a dramatic increase in size and number of splenic melanomacrophage centres, suggesting increased levels of the hemosiderin (resulting from the erythrocyte's destruction) and lipofuscin, common in tissue catabolism and degenerative chronic disorders[14][15]. Secondary infections with other parasites and bacteria are common for the S. chrysophrii-infected seabream[16].
Impact
In the experimental studies, fingerling seabream (30 g) had prevalence up to the 100%, and the lager fish (150 g) 96.6%[10][3]. Some authors found positive correlation between increase of sexual hormone levels and susceptibility to monogeneans[17][18], while others suggest that in the larger fish, water current passing through the gills is stronger than in the smaller fish, therefore disabling the settlement of oncomiracidia[10].
Diagnosis
S. chrysophrii infection is diagnosed by examining the gills under the stereomicroscope and determining the presence of the different parasite stages.
Treatments
Currently, only formalin baths are regulated in some European countries. 30 min bath in formalin (300 ppm) is 100% effective for eggs, oncomiracidia and adults in in vitro treatments, and hydrogen peroxide (200 ppm) is 100% effective for oncomiracidia and adults[9].
Other control strategies
Recommend synchronization of fish baths with net changing to lower the possibility of infection or re-infection[9]. Regular parasite counts from gills are recommended to decide bath treatments and avoid sudden outbreaks.
Research
Different aspects of monogenean biology, ecology and pathology have been investigated through the EU funded Horizon2020 Project ParaFishControl, adding greatly to the generation of new fundamental and applicable knowledge. The genome and transcriptome of S. chrysophrii different developmental stages has been sequenced; the gilt-head seabream response to chronic and mild infection by S. chrysophrii has been evaluated using RNA-seq, as well standard histology and immunohistochemistry techniques; an array of synthetic compounds and herbal extracts has been tested to infer toxicity on the adult monogenean; assessment of the transfer of the monogenean between wild and farmed fish has been done using ddRAD-seq; different techniques of in vitro culture have been successfully applied.

References
- Van Beneden, P. J., & Hesse, C. E. (1863). Second Appendice au Mémoire sur les Bdellodes et les Trématodes: Mémoire présenté à l'Académie Royale de Belgique.
- Mamaev, Y. L. (1984). [The composition of the genera Atriaster and Atrispinum (Microcotylidae, Monogenea) and some peculiarities of their morphology]. Parazitologiya, 18(3), 204-208. [in Russian]
- Antonelli, Laetitia; Quilichini, Yann; Marchand, Bernard (2010). "Sparicotyle chrysophrii (Van Beneden and Hesse 1863) (Monogenea: Polyopisthocotylea) parasite of cultured Gilthead sea bream Sparus aurata (Linnaeus 1758) (Pisces: Teleostei) from Corsica: ecological and morphological study". Parasitology Research. 107 (2): 389–398. doi:10.1007/s00436-010-1876-0. ISSN 0932-0113. PMID 20422218.
- Euzet, L., C. Combes. 1998. The selection of habitats among the monogenea. Int. J. Parasitol., 28: 1645-1652.
- Sanz, F. 1992. Mortality of cultured seabream (Sparus aurata) caused by an infection with a trematode of the genus Microcotyle. Bull. Eur. Assoc. Fish Pathol. 12:186–188.
- Alvarez-Pellitero, P. 2004. Report about fish parasitic diseases. Alvarez-Pellitero P, Barja JL, Basurco B, Berthe F, Toranzo AE (Eds). Mediterranean Aquaculture Diagnostic Laboratories 103-129.
- Vagianou, S., Athanassopoulou, F., Ragias, V., Di Cave, D., Leontides, L., Golomazou, E. 2006. Prevalence and pathology of ectoparasites of Mediterranean Sea bream and sea bass reared under different environmental and aquaculture conditions. Isr. J. Aquacult. Bamidgeh 58: 78-88.
- Repullés-Albelda, A., Holzer, A.S., Raga, J.A., Montero, F.E. 2012. Oncomiracidial development, survival and swimming behaviour of the monogenean Sparicotyle chrysophrii (Van Beneden and Hesse, 1863). Aquaculture, 338, pp.47-55.
- Sitjà-Bobadilla, A., de Felipe, M.C., Alvarez-Pellitero, P. 2006. In vivo and in vitro treatments against Sparicotyle chrysophrii (Monogenea: Microcotylidae) parasitizing the gills of gilthead seabream (Sparus aurata L.). Aquaculture 261:856–864.
- Sitjà-Bobadilla, A., Alvarez-Pellitero, P. 2009. Experimental transmission of Sparicotyle chrysophrii (Monogenea: Polyopisthocotylea) to gilthead seabream (Sparus aurata) and histopathology of the infection. Folia Parasitol. 56:143–151.
- Roubal, F.R. 1994. Attachment of eggs by Lamellodiscus acanthopagri (Monogenea: Diplectanidae) to the gills of Acanthopagrus australis (Pisces: Sparidae), with evidence for auto-infection and postsettlement migration. Can. J. Zool. 72:87–95.
- Mahmoud, N.E., Mahmoud, A.M., Fahmy, M.M. 2014. Parasitological and Comparative Pathological Studies on Monogenean Infestation of Cultured Sea Bream (Sparus aurata, Spariidae) in Egypt. Oceanography, 2(4), p.1000129.
- De Vico, G., Cataldi, M., Carella, F., Marino, F., Passantino, A. 2008. Histological, histochemical and morphometric changes of splenic melanomacrophage centers (SMMCs) in Sparicotyle infected cultured sea breams (Sparus aurata). - Immunopharmacology and immunotoxicology, 30: 27-35.
- Agius, C., Roberts, R.J. 1981. Effects of starvation on the melano-macrophage centres of fish. Fish. Soc. Brit. Isles, 19, 161–169.
- Agius, C., Agbede, S.A. 1984. An electron microscopical study on the genesis of lipofuscin, melanin and haemosiderin in the haemopoietic tissues of fish. Fish. Soc. Brit. Isles., 24, 471–488.
- Cruz e Silva, M. P., Grazina Freitas, M. S., Orge, M. L. 1997. Co-Infection by Monogenetic Trematodes of the Genus Microcotyle V. BENEDEN & HESSE, 1863, Lamellodiscus ignoratus PALOMBI, 1943, the Protozoan Trichodina sp. EHNRENBERG, 1838, and the presence of Epitheliocystis, Vibrio algynoliticus and V. vulnificus in cultured Seabream (Sparus aurata L.) in Portugal. Bull. Eur. Ass. Fish Pathol. 17: 40-42.
- Buchmann, K., 1997. Population increase of Gyrodactylus derjavini on rainbow trout induced by testosterone treatment of the host. Dis. Aquat. Organ. 30:145–150.
- Escobedo, G., Roberts, C.W., Carrero, J.C., Morales-Montor, J. 2005. Parasite regulation by host hormones: an old mechanism of host exploitation? Trends Parasitol. 21:588–593