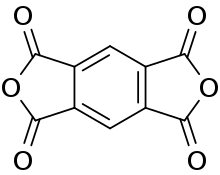
Pyromellitic dianhydride
Pyromellitic dianhydride (PMDA) is an organic compound with the formula C6H2(C2O3)2. It is the double carboxylic acid anhydride that is used in the preparation of polyimide polymers such as Kapton. It is a white solid.
 | |
| Identifiers | |
|---|---|
| ECHA InfoCard | 100.001.726 |
| UNII | |
CompTox Dashboard (EPA) |
|
| Properties | |
| C10H2O6 | |
| Molar mass | 218.120 g·mol−1 |
| Appearance | White solid |
| Density | 1.68 g/cm3 |
| Melting point | 283 to 286 °C (541 to 547 °F; 556 to 559 K) |
| Boiling point | 397 to 400 °C (747 to 752 °F; 670 to 673 K) |
| Hygroscopic | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
It is prepared by gas-phase oxidation of 1,2,4,5-tetramethylbenzene (or related tetrasubstituted benzene derivatives). An idealized equation is:[1]
- C6H2(CH3)4 + 6 O2 → C6H2(C2O3)2 + 6 H2O
In the laboratory, it can be prepared by dehydration of pyromellitic acid using acetic anhydride.[2]
Reactions
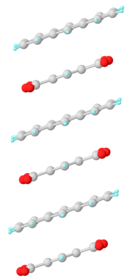
Structure of the complex formed upon co-crystallization of pyromellitic anhydride (molecules terminated in red) and anthracene.[3]
PMDA is an electron-acceptor, forming a variety of charge-transfer complexes. It reacts with amines to diimides, C6H2[(CO)2NR]2 which also have acceptor properties.[4]
Safety
Some evidence suggests that PMDA causes occupational asthma.[5]
gollark: Great, my hatchery has removed the adults fine.
gollark: *is always locked, except now*
gollark: Should I just put up my hatchery on the forum now to try and get more feedback/views or wait a bit?
gollark: DR is filled up for the next eternity.
gollark: I suggested procedurally generated nebulae. On Bad Ideas.
References
- F. Röhrscheid (2012). "Carboxylic Acids, Aromatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_249.
- E. Philippi, R. Thelen. (1930). "Pyromellitic Acid". Organic Syntheses. 10: 90. doi:10.15227/orgsyn.010.0090.
- Robertson, B. E.; Stezowski, J. J. (1978). "The crystal structure of the π-molecular complex of anthracene with pyromellitic dianhydride at –120°C". Acta Crystallographica Section B. 34 (10): 3005–3011. doi:10.1107/S0567740878009929.
- Song, Zhiping; Zhan, Hui; Zhou, Yunhong (2010). "Polyimides: Promising Energy-Storage Materials". Angewandte Chemie International Edition. 49 (45): 8444–8448. doi:10.1002/anie.201002439. PMID 20862664.CS1 maint: multiple names: authors list (link)
- Madsen, Milene Torp; Skadhauge, Lars Rauff; Nielsen, Anders Daldorph; Baelum, Jesper; Sherson, David Lee (2019). "Pyromellitic dianhydride (PMDA) may cause occupational asthma". Occupational and Environmental Medicine. 76 (3): 175–177. doi:10.1136/oemed-2018-105295. PMC 6581108. PMID 30635433.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.