Psymberin
Psymberin, also known as irciniastatin A, is a cytotoxin derived from sea sponges. It was discovered by two independent research groups in 2004.[1][2] Psymberin was found to be highly bioactive as it showed LC50s at nanomolar concentrations against various types of tumors.[1][2]
 | |
| Names | |
|---|---|
| IUPAC name
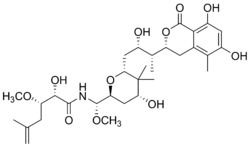
(2S,3S)-N-((S)-((2S,4R,6R)-6-((2S,3R)-3-((R)-6,8-dihydroxy-5-methyl-1-oxoisochroman-3-yl)-2-hydroxybutyl)-4-hydroxy-5,5-dimethyltetrahydro-2H-pyran-2-yl)(methoxy)methyl)-2-hydroxy-3-methoxy-5-methylhex-5-enamide | |
| Other names
irciniastatin A | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| |
| Properties | |
| C31H47NO11 | |
| Molar mass | 609.71 g/mol |
| Appearance | clear, glasslike solid[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Biosynthesis
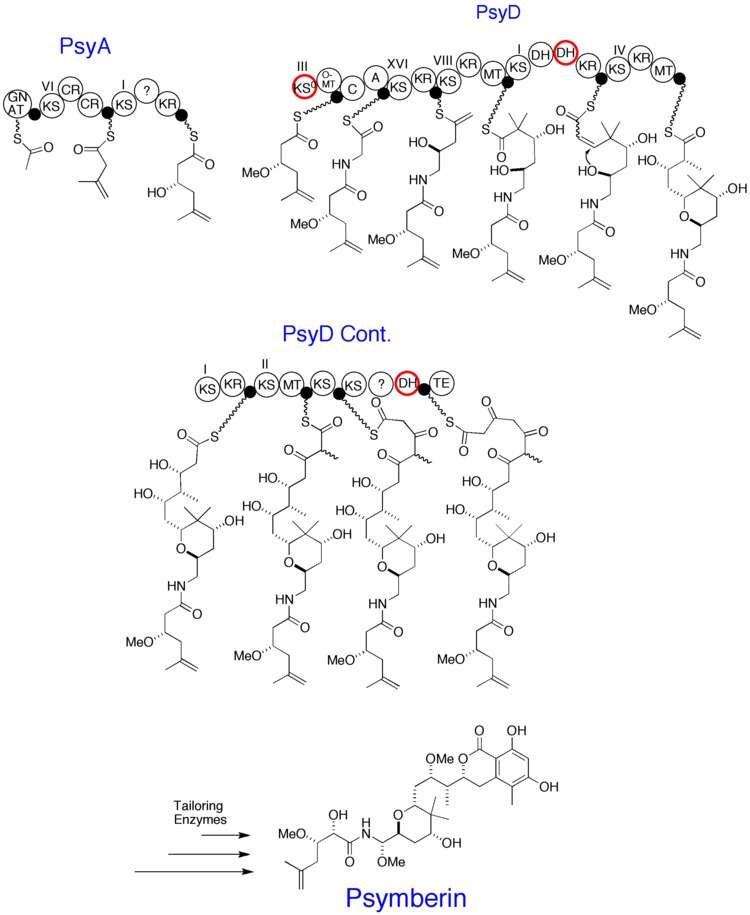
The biosynthesis of psymberin became of interest when it was found to have a very similar structure to pederin, a natural product derived from the beetle genus Paederus.[3] This led researchers to investigate the genes of the organism that code for the respective natural product. A trans-AT polyketide synthase cluster, designated as the Psy cluster, contained two large groups of genes coding for two PKSs.[3] The smaller gene group, psyA, encoded a PKS with three modules, while the larger psyD encoded a ten module PKS. This ten module PsyD protein is, to date, the protein with the highest reported number of PKS modules.[3] Large portions of the gene cluster psyD have very close counterparts in the pederin gene clusters. This indicates that psymberin and pederin are both structurally and biosynthetically related. The figure below shows the proposed biosynthesis of psymberin based on analysis of the gene clusters. Roman numerals represent which clade each Ketosynthase domain belongs. The domains outlined in red represent carrier proteins. The following abbreviations correspond to the figure below: GNAT, GCN5-related N-acetyltransferase; CR, crotonase; KR, ketoreductase; KS0, non-elongating ketoreductase; OMT, O-methyltranfserase; C, nonribosomal peptide synthetase (NRPS) condensation domain; A, NRPS adenylation domain; MT, Methyltransferase; DH, deydratase; TE, Thioesterase; ?, unknown.
See also
References
- Cichewicz RH, Valeriote FA, Crews P (2004). "Psymberin, a potent sponge-derived cytotoxin from Psammocinia distantly related to the pederin family". Org. Lett. 6 (12): 1951–4. doi:10.1021/ol049503q. PMID 15176791.
- Pettit GR, Xu JP, Chapuis JC, Pettit RK, Tackett LP, Doubek DL, Hooper JN, Schmidt JM (2004). "Antineoplastic agents. 520. Isolation and structure of irciniastatins A and B from the Indo-Pacific marine sponge Ircinia ramosa". J. Med. Chem. 47 (5): 1149–52. doi:10.1021/jm030207d. PMID 14971894.
- Fisch KM, Gurgui C, Heycke N, van der Sar SA, Anderson SA, Webb VL, Taudien S, Platzer M, Rubio BK, Robinson SJ, Crews P, Piel J (2009). "Polyketide assembly lines of uncultivated sponge symbionts from structure-based gene targeting". Nat. Chem. Biol. 5 (7): 494–501. doi:10.1038/nchembio.176. PMID 19448639.
External links
- Psymberin at the US National Library of Medicine Medical Subject Headings (MeSH)