Pentaamine(dinitrogen)ruthenium(II) chloride
Pentaamine(nitrogen)ruthenium(II) chloride is an inorganic compound with the formula [Ru(NH3)5(N2)]Cl2. It is a nearly white solid, but its solutions are yellow. The cationic complex is of historic significance as the first compound with N2 bound to a metal center.[1][2] [Ru(NH3)5(N2)]2+ adopts an octahedral structure with C4v symmetry.[3]
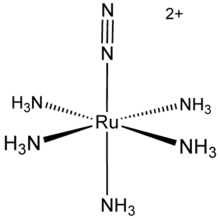 | |
| Names | |
|---|---|
| IUPAC name
pentaamminedinitrogenruthenium (II) chloride | |
| Identifiers | |
3D model (JSmol) |
|
| |
| Properties | |
| H15N7Cl2Ru | |
| Molar mass | 285.14 g/mol |
| Appearance | colorless solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation and properties
Pentaamine(nitrogen)ruthenium(II) chloride is synthesized in an aqueous solution from pentaamminechlororuthenium(III) chloride, sodium azide, and methanesulfonic acid:[4]
- [Ru(NH3)5Cl]Cl2 + NaN3 → [Ru(NH3)5N2]Cl2 + ...
If it is to be used in situ, the cation can be made more conveniently from ruthenium(III) chloride and hydrazine hydrate:[4]
- RuCl3 + 4 N2H4 → [Ru(NH3)5N2]2+ + ...
This N2 complex is stable in aqueous solution and has a relatively low ligand exchange rate with water. Being a d6 complex, the Ru-N bond is stabilized by the pi backbonding, the donation of metal d-electrons into the N2 π* orbitals.[5] The related metal ammine complex [Os(NH3)5(N2)]2+ is also known.
Reactions
The dinitrogen ligand is not reduced by aqueous sodium borohydride.[6] Nearly all known reactions of this compound are displacement reactions. Pentaamine(halogen)ruthenium(II) halides can be synthesized by treating [Ru(NH3)5N2]2+ with halide sources:[4]
- [Ru(NH3)5N2]2+ + X− → [Ru(NH3)5X]+ + N2
[Ru(NH3)5N2]2+ forms the symmetrically bridging symmetrical dinitrogen complex [(NH3)5Ru-NN-Ru(NH3)5]4+.[7][8]
References
- Alen. A. D.; Senoff, C. V. (1965) "Nitrogenopentammineruthenium(II) complexes" Chemical Communications 24: 621-2. doi:10.1039/C19650000621
- Senoff, C.V. (1990) "The discovery of [Ru(NH3)5NN2]2+: A case of serendipity and the scientific method" Journal of Chemical Education, volume 67: 368. doi:10.1021/ed067p368
- Fergusson, J. E.; Love, J. L.; Robinson, W. T. (1972) "The Crystal and Molecular Structure of Dinitrogenpentaammineosmium(II) Chloride, [Os(NH3)5N2]Cl2, and Related Ruthenium Complexes" Inorg. Chem., 11, pp. 1662–1666. doi:10.1021/ic50113a042
- Allen, A. D., Bottomley, F., Harris, R. O., Reinsalu, V. P., Senoff, C. V. "Pentaammine(Nitrogen)Ruthenium(II) Salts and Other Ammines of Ruthenium" Inorganic Syntheses, 2007, volume 12, pp. 2-8. doi:10.1002/9780470132432.ch1
- Taube, H. "Chemistry of Ruthenium(II) and Osmium(II) Ammines" Pure and Applied Chemistry, 1979, volume 51, p. 901–12. doi:10.1351/pac197951040901
- J. Chatt, R. L. Richards, J. F. Fergusson and J. L. Love, "The reduction of nitrogen complexes" Chem. Commun. 1968, 1522. doi:10.1039/C19680001522
- D. F. Harrison, F. Weissberger and H. Taube, Science, 1968, volume 159, 320.
- Chatt, J. "Molecular Nitrogen as a Ligand" Pure Appl. Chem., 1970, volume 24, pp. 425–442. doi:10.1351/pac197024020425