PDIA2
Protein disulfide isomerase family A member 2 is a protein that in humans is encoded by the PDIA2 gene. [5]
Function
This gene encodes a member of the disulfide isomerase (PDI) family of endoplasmic reticulum (ER) proteins that catalyze protein folding and thiol-disulfide interchange reactions. The encoded protein has an N-terminal ER-signal sequence, two catalytically active thioredoxin (TRX) domains, two TRX-like domains and a C-terminal ER-retention sequence. The protein plays a role in the folding of nascent proteins in the endoplasmic reticulum by forming disulfide bonds through its thiol isomerase, oxidase, and reductase activity. The encoded protein also possesses estradiol-binding activity and can modulate intracellular estradiol levels. [provided by RefSeq, Sep 2017].
References
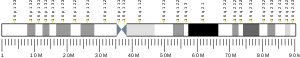
- GRCh38: Ensembl release 89: ENSG00000185615 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000024184 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Entrez Gene: Protein disulfide isomerase family A member 2". Retrieved 2017-12-20.
Further reading
- VanderWaal RP, Spitz DR, Griffith CL, Higashikubo R, Roti Roti JL (2002). "Evidence that protein disulfide isomerase (PDI) is involved in DNA-nuclear matrix anchoring". J. Cell. Biochem. 85 (4): 689–702. doi:10.1002/jcb.10169. PMID 11968009.
- Ko HS, Uehara T, Nomura Y (2002). "Role of ubiquilin associated with protein-disulfide isomerase in the endoplasmic reticulum in stress-induced apoptotic cell death". J. Biol. Chem. 277 (38): 35386–92. doi:10.1074/jbc.M203412200. PMID 12095988.
- Lumb RA, Bulleid NJ (2002). "Is protein disulfide isomerase a redox-dependent molecular chaperone?". EMBO J. 21 (24): 6763–70. doi:10.1093/emboj/cdf685. PMC 139105. PMID 12485997.
- Clissold PM, Bicknell R (2003). "The thioredoxin-like fold: hidden domains in protein disulfide isomerases and other chaperone proteins". BioEssays. 25 (6): 603–11. doi:10.1002/bies.10287. PMID 12766950.
- Pirneskoski A, Klappa P, Lobell M, Williamson RA, Byrne L, Alanen HI, Salo KE, Kivirikko KI, Freedman RB, Ruddock LW (2004). "Molecular characterization of the principal substrate binding site of the ubiquitous folding catalyst protein disulfide isomerase". J. Biol. Chem. 279 (11): 10374–81. doi:10.1074/jbc.M312193200. PMID 14684740.
- Spooner RA, Watson PD, Marsden CJ, Smith DC, Moore KA, Cook JP, Lord JM, Roberts LM (2004). "Protein disulphide-isomerase reduces ricin to its A and B chains in the endoplasmic reticulum". Biochem. J. 383 (Pt 2): 285–93. doi:10.1042/BJ20040742. PMC 1134069. PMID 15225124.
- Li SJ, Hong XG, Shi YY, Li H, Wang CC (2006). "Annular arrangement and collaborative actions of four domains of protein-disulfide isomerase: a small angle X-ray scattering study in solution". J. Biol. Chem. 281 (10): 6581–8. doi:10.1074/jbc.M508422200. PMID 16407203.
- Otsu M, Bertoli G, Fagioli C, Guerini-Rocco E, Nerini-Molteni S, Ruffato E, Sitia R (2006). "Dynamic retention of Ero1alpha and Ero1beta in the endoplasmic reticulum by interactions with PDI and ERp44". Antioxid. Redox Signal. 8 (3–4): 274–82. doi:10.1089/ars.2006.8.274. PMID 16677073.
- Park B, Lee S, Kim E, Cho K, Riddell SR, Cho S, Ahn K (2006). "Redox regulation facilitates optimal peptide selection by MHC class I during antigen processing". Cell. 127 (2): 369–82. doi:10.1016/j.cell.2006.08.041. PMID 17055437.
This article incorporates text from the United States National Library of Medicine, which is in the public domain.