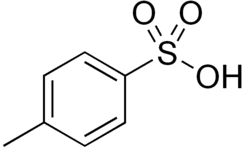
p-Toluenesulfonic acid
p-Toluenesulfonic acid (PTSA or pTsOH) or tosylic acid (TsOH) is an organic compound with the formula CH3C6H4SO3H. It is a white solid that is soluble in water, alcohols, and other polar organic solvents. The CH3C6H4SO2– group is known as the tosyl group and is often abbreviated as Ts or Tos. Most often, TsOH refers to the monohydrate, TsOH.H2O.
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
4-Methylbenzene-1-sulfonic acid | |||
| Other names
4-Methylbenzenesulfonic acid Tosylic acid tosic acid PTSA | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.002.891 | ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C7H8O3S | |||
| Molar mass | 172.20 g/mol (anhydrous) 190.22 g/mol (monohydrate) | ||
| Appearance | colorless (white) solid | ||
| Density | 1.24 g/cm3 | ||
| Melting point | 38 °C (100 °F; 311 K) (anhydrous)[2] 103 to 106 °C (217 to 223 °F; 376 to 379 K) (monohydrate) | ||
| Boiling point | 140 °C (284 °F; 413 K) at 20 mmHg | ||
| 67 g/100 mL | |||
| Acidity (pKa) | −2.8 (water) reference for benzenesulfonic acid,[3] 8.5 (acetonitrile)[4] | ||
| Structure | |||
| tetrahedral at S | |||
| Hazards | |||
| Main hazards | skin irritant | ||
| Safety data sheet | External MSDS | ||
| R-phrases (outdated) | R36/37/38 | ||
| S-phrases (outdated) | S26 | ||
| Related compounds | |||
Related sulfonic acids |
Benzenesulfonic acid Sulfanilic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
As with other sulfonic acids, TsOH is a strong organic acid. It is about one million times stronger than benzoic acid. It is one of the few strong acids that is solid and, hence, conveniently weighed.
Preparation and handling
TsOH is prepared on an industrial scale by the sulfonation of toluene. It hydrates readily. Common impurities include benzenesulfonic acid and sulfuric acid. Monohydrate pTSA contains crystalline water as well as water as Impurity. To estimate the total moisture present as impurity, Karl Fischer method is used. Impurities can be removed by recrystallization from its concentrated aqueous solution followed by azeotropic drying with toluene.[5]
TsOH finds use in organic synthesis as an "organic-soluble" acid catalyst. Examples of uses include:
- Acetalization of an aldehyde.[6]
- Esterification of carboxylic acids.[7]
- Transesterification of an ester.[8]
Tosylates
Alkyl tosylates are alkylating agents because tosylate is electron-withdrawing, which makes the tosylate anion a good leaving group. Tosylate is a pseudohalide. Toluenesulfonate esters undergo nucleophilic attack or elimination. Reduction of tosylate esters gives the hydrocarbon. Thus, tosylation followed by reduction allows for the deoxygenation of alcohols.
In a famous and illustrative use of tosylate, 2-norbornyl cation was displaced from the 7-norbornenyl tosylate. The elimination occurs 1011 faster than the solvolysis of anti-7-norbornyl p-toluenesulfonate.[9]
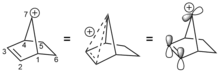
Tosylates are also protecting group for alcohols. They are prepared by combining the alcohol with 4-toluenesulfonyl chloride, usually in an aprotic solvent, often pyridine.[10]
Reactions
- TsOH may be converted to p-toluenesulfonic anhydride by heating with phosphorus pentoxide.[11]
- When heated with acid and water, TsOH undergoes hydrolysis to toluene:
- CH3C6H4SO3H + H2O → C6H5CH3 + H2SO4
This reaction is general for aryl sulfonic acids.[12][13]
See also
References
- Merck Index, 11th Edition, 9459.
- Armarego, Wilfred (2003). Purification of Laboratory Chemicals. Elsevier Science. p. 370. ISBN 0-7506-7571-3.
- Guthrie, J. P. Hydrolysis of esters of oxy acids: pKa values for strong acids. Can. J. Chem. 1978, 56, 2342-2354.
- Eckert, F.; Leito, I.; Kaljurand, I.; Kütt, A.; Klamt, A.; Diedenhofen, M. Prediction of Acidity in Acetonitrile Solution with COSMO-RS. J. Comput. Chem. 2009, 30, 799-810. doi:10.1002/jcc.21103
- Perrin, D. D. & Armarego, W. L. F. (1988). Purification of Laboratory Chemicals. Oxford: Pergamon Press.
- H. Griesser, H.; Öhrlein, R.; Schwab, W.; Ehrler, R.; Jäger, V. (2004). "3-Nitropropanal, 3-Nitropropanol, and 3-Nitropropanal Dimethyl Acetal". Organic Syntheses.CS1 maint: multiple names: authors list (link); Collective Volume, 10, p. 577
- Furuta, K. Gao, Q.-z.; Yamamoto, H. (1998). "Chiral (Acyloxy)borane Complex-catalyzed Asymmetric Diels-Alder Reaction: (1R)-1,3,4-Trimethyl-3-cyclohexene-1-carboxaldehyde". Organic Syntheses.CS1 maint: multiple names: authors list (link); Collective Volume, 9, p. 722
- Imwinkelried, R.; Schiess, M.; Seebach, D. (1993). "Diisopropyl (2S,3S)-2,3-O-isopropylidenetartrate". Organic Syntheses.CS1 maint: multiple names: authors list (link); Collective Volume, 8, p. 201
- Winstein, S.; Shatavsky, M.; Norton, C.; Woodward, R. B. (1955-08-01). "7-Norbornenyl and 7-Norbornyl cations". Journal of the American Chemical Society. 77 (15): 4183–4184. doi:10.1021/ja01620a078. ISSN 0002-7863.
- "Nucleophilic Substitution".
- L. Field and J. W. McFarland (1963). "p-Toluenesulfonic Anhydride". Organic Syntheses.; Collective Volume, 4, p. 940
- C. M. Suter (1944). The Organic Chemistry of Sulfur. New York: John Wiley & Sons. pp. 387–388.
- J. M. Crafts (1901). "Catalysis in concentrated solutions". J. Am. Chem. Soc. 23: 236–249.