Osazone
Osazones are a class of carbohydrate derivatives found in organic chemistry formed when reducing sugars are reacted with excess of phenylhydrazine at boiling temperatures.[1] The osazone formation reaction was developed by famous German chemist Emil Fischer,[2] who used the reaction as a test to identify monosaccharides whose stereochemistry differed by only one chiral carbon.[3][4]
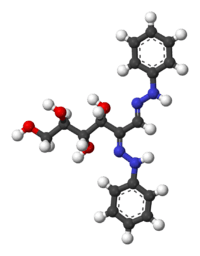
The reaction involves formation of a pair of phenylhydrazone functionalities, concomitant with the oxidation of the hydroxy group on the alpha carbon (carbon atom adjacent to the carbonyl center). This formation allows two sugars with closely related structures to give the same osazone. Glucosazone and fructosazone, for example, are identical.[5] Since the reaction requires a free carbonyl group, only reducing sugars can form osazones. Sucrose, for example, is unable to form an osazone, because it is non-reducing.
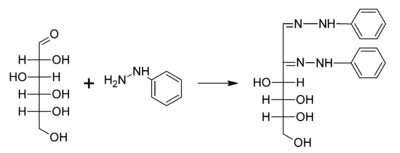 A typical reaction showing the formation of an osazone. D-glucose reacts with phenylhydrazine to give glucosazone. The same product is obtained from fructose and mannose.
A typical reaction showing the formation of an osazone. D-glucose reacts with phenylhydrazine to give glucosazone. The same product is obtained from fructose and mannose.
The mechanism involves two steps, each of which forms one of the phenylhydrazone functionalities. In the first step, a molecule of phenylhydrazine reacts with the carbonyl group on the sugar to form a phenylhydrazone by elimination of a water molecule. The next step involves two molecules of phenylhydrazine. The first oxidizes the hydroxy group on the reactive alpha carbon to a carbonyl group, and the second then forms a hydrazone with it.[6] The end product is stabilized by a hydrogen bond between the two phenylhydrazone groups.[1]
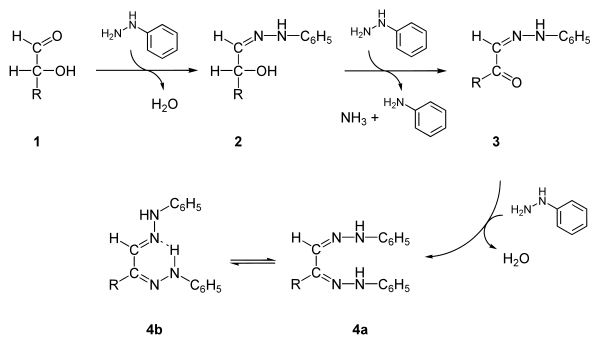 General steps in osazone formation
General steps in osazone formation
Osazones are highly coloured and crystalline compounds and can be easily detected. Each different osazone has a characteristic crystal formation.[7]
References
- Mester, L.; El Khadem, H.; Horton, D. (1970). "Structure of saccharide osazones". Journal of the Chemical Society C: Organic (18): 2567. doi:10.1039/J39700002567.
- Helferich, B. (1953). "Emil Fischer zum 100. Geburtstag". Angewandte Chemie. 65 (2): 45–52. doi:10.1002/ange.19530650202.
- Fischer, Emil (1908). "Schmelzpunkt des Phenylhydrazins und einiger Osazone". Berichte der Deutschen Chemischen Gesellschaft. 41: 73–77. doi:10.1002/cber.19080410120.
- Fischer, Emil (1894). "Ueber einige Osazone und Hydrazone der Zuckergruppe". Berichte der Deutschen Chemischen Gesellschaft. 27 (2): 2486–2492. doi:10.1002/cber.189402702249.
- Ramakrishnan, S. (2004). Textbook of Medical Biochemistry. Orient Blackswan. ISBN 9788125020714.
- Barry, VINCENT C.; Mitchell, PW (1955). "Mechanism of Osazone Formation". Nature. 175 (4448): 220. Bibcode:1955Natur.175..220B. doi:10.1038/175220a0. PMID 13235861.
- Gupta, Anil (2019). "Carbohydrates". Comprehensive Biochemistry for Dentistry. Singapore: Springer. pp. 108–110. ISBN 978-981-13-1035-5.