Moniliformin
Moniliformin is an unusual mycotoxin, a feed contaminant that is lethal to fowl, especially ducklings.
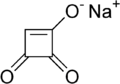 | |
| Names | |
|---|---|
| IUPAC name
sodium 3,4-dioxo-1-cyclobutenolate | |
| Other names
Semisquaric acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4HNaO3 | |
| Molar mass | 120.04 g/mol |
| Appearance | Yellow crystalline solid |
| Melting point | Decomposes at 345-355 °C without melting |
| very good | |
| Related compounds | |
Related compounds |
Squaric acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Moniliformin is formed in many cereals by a number of Fusarium species that include Fusarium moniliforme, Fusarium avenaceum, Fusarium subglutinans, Fusarium proliferatum, Fusarium fujikuroi and others. It is mainly cardiotoxic and causes ventricular hypertrophy. Moniliformin actually causes competitive inhibition of the activity of pyruvate dehydrogenase complex of respiratory reaction, which prevents pyruvic acid, product of glycolysis, to convert to acetyl CoA.[1][2][3] Ultrastructural examination of right ventricular wall of 9 month old female mink (Mustela vison) fed acute doses of moniliformin (2.2 and 2.8 mg/kg diet) and sub-acute doses (1.5 to 3.2 mg/kg diet) reveals significant damage to myofiber, mitochondria, Z and M lines and sarcoplasmic reticulum as well as increased extracellular collagen deposition. Mink is considered most sensitive mammals to the toxicity of moniliformin.[4] Chemically speaking, it is the sodium salt of deoxysquaric acid
Physicochemical information
IUPAC name: 3-hydroxy-3-cyclobutene-1,2-dione. Solubility information: Moniliformin is soluble in water and polar solvents, such as methanol.
λmax: 226, 259 in methanol
See also
Sources and references
- Thiel, Pieter G (1978). "A molecular mechanism for the toxic action of moniliformin, a mycotoxin produced by fusarium moniliforme". Biochemical Pharmacology. 27 (4): 483–6. doi:10.1016/0006-2952(78)90381-7. PMID 629807.
- Moniliformin product page from Fermentek
- Moniliformin information leaflet by Romerlab
- Morgan MK, Fitzgerald SD, Rottinghaus GE, Bursian SJ and Aulerich RJ. 1999. Toxic effects to mink of moniliformin extracted from Fusarium fujikuroi culture material. Veterinary and Human Toxicology 1(1):pp-1-5