Methyltriphenylphosphonium bromide
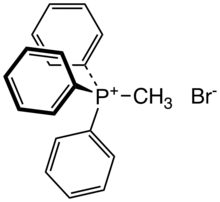
Methyltriphenylphosphonium bromide is the organophosphorus compound with the formula [(C6H5)3PCH3]Br. It is the bromide salt of a phosphonium cation. It is a white salt that is soluble in polar organic solvents.
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C19H18BrP | |
| Molar mass | 357.231 g·mol−1 |
| Appearance | white solid |
| Hazards | |
| GHS pictograms |    |
| GHS Signal word | Danger |
GHS hazard statements |
H300, H301, H302, H312, H315, H319, H332, H411 |
| P261, P264, P270, P271, P273, P280, P301+310, P301+312, P302+352, P304+312, P304+340, P305+351+338, P312, P321, P322, P330, P332+313, P337+313, P362, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis and reactions
Methyltriphenylphosphonium bromide is produced by treating triphenylphosphine with methyl bromide:[1]
- Ph3P + CH3Br → Ph3PCH3Br
Methyltriphenylphosphonium bromide is the principal precursor to methylenetriphenylphosphorane, a useful methylenating reagent. This conversion is achieved by treating methyltriphenylphosphonium bromide with strong base.[2]
- Ph3PCH3Br + BuLi → Ph3PCH2 + LiBr + BuH
gollark: Anyway, you hold shift to boost in the direction you look, and it has limited anti-fall-damage capability.
gollark: It vanished mysteriously and I got refunded.
gollark: I bought it for 100KST.
gollark: I used to have a command computer.
gollark: I do have a somewhat usable non-elytra flight thing.
References
- Wittig, Georg; Schoellkopf, U. (1960). "Methylenecyclohexane". Organic Syntheses. 40: 66. doi:10.15227/orgsyn.040.0066.
- Fitjer, Lutz; Quabeck, Ulrike (1985). "The Wittig Reaction Using Potassium-tert-butoxide High Yield Methylenations of Sterically Hindered Ketones". Synthetic Communications. 15 (10): 855–864. doi:10.1080/00397918508063883.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.