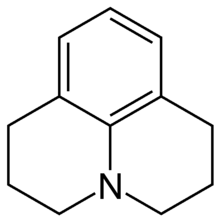
Julolidine
Julolidine is a heterocyclic aromatic organic compound. It has the formula C12H15N.
 | |
| Names | |
|---|---|
| IUPAC name
Julolidine | |
| Other names
2,3,6,7-tetrahydro-1H,5H-benzo[ij]quinolizine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.006.851 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H15N | |
| Molar mass | 173.259 g·mol−1 |
| Density | 1.003 g/mL |
| Melting point | 35 °C (95 °F; 308 K) |
Refractive index (nD) |
1.568 |
| Hazards | |
| Flash point | 110 °C (230 °F; 383 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis
The first synthesis of julolidine was first reported by G. Pinkus in 1892.[1]
Applications
This compound and its derivatives have found recent interest as photoconductive materials, chemiluminescence substances, chromogenic substrates in analytical redox reactions, dye intermediates, potential antidepressants and tranquilizers, nonlinear optical materials, high sensitivity photopolymerizable materials, and for improving color stability in photography.
gollark: Cruelty is what we do best at PotatOS Network Systems™.
gollark: Maybe have a *fake* goal, but it's not real and is fake.
gollark: Oh, maybe you can randomly win the lottery or something, but it's entirely random and extremely rare. And it doesn't actually fix all your problems.
gollark: You can't *win* life, why should petcruel be different?
gollark: I think this came from a discussion about helloboi's virtual pet game.
References
- Pinkus, G. Ueber die Einwirkung von Trimethylenchlorbromid auf einige aromatische Amine und Amide. Berichte der deutschen chemischen Gesellschaft 1892, 25 (2), 2798–2806
External links
- Katritzky, Alan R.; Rachwal, Bogumila; Rachwal, Stanislaw; Abboud, Khalil A. (1996). "Convenient Synthesis of Julolidines Using Benzotriazole Methodology". The Journal of Organic Chemistry. 61 (9): 3117. doi:10.1021/jo9519118. PMID 11667174.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.