Gregarina garnhami
Gregarina garnhami is a eukaryotic unicellular organism belonging to the Apicomplexa described in 1956 by Canning as a parasite found in several locusts, such as the desert locust, African migratory locust, and Egyptian locust. Especially, the desert locust is the host for this species, as up to 100% of animals can become infected.[1] An estimated thousands of different species of gregarines can be in insects and 99% of these gregarines still need to be described. Each insect is said to host multiple species.[2][3] A remarkable feature of G. garnhami is its autofluorescence.
| Gregarina garnhami | |
|---|---|
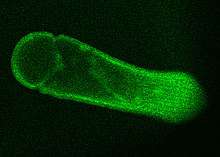 | |
| Autofluorescence of Gregarina garnhami | |
| Scientific classification | |
| Domain: | |
| (unranked): | |
| (unranked): | |
| Phylum: | |
| Class: | |
| Subclass: | |
| Order: | |
| Family: | Gregarinidae |
| Genus: | |
| Species: | G. garnhami |
| Binomial name | |
| Gregarina garnhami Canning 1956[1] | |
Cell structure
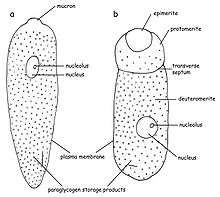
G. garnhami belongs to the septate eugregarines, meaning its cell is separated into parts. In G. garnhami, three parts can be seen: epimerite, protomerite, and deutomerite, but their visibility depends on the lifestage of the organism. A characteristic of gregarins is the typical construction of the pellicula that is formed by a cell membrane and two cytoplasmamembranes (the latter is often referred to as the inner membrane complex, IMC or alveoli). The membranes' proximity to one another often makes them difficult to distinguish. This structure is often also referred to as the trilayered structure. Beneath the inner membrane, a basal (internal) lamina can be seen. This basal lamina separates the epicytic foldings from the rest of the cell. These epicytic foldings form the outer surface of G. garnhami, and hundreds of these can be observed at the surface enlarging the surface of the cell.[4][5][6][7][8] The cytoplasm of the cell (in the epimerite, protomerite, and deutomerite) is divided in two zones: ectoplasm and endoplasm. The ectoplasm is clear and does not contain much granular material. In the ectoplasm, the microtubules can be found. The endoplasm is less transparent and contains paraglycogen giving the cells a brown-yellowish color when viewing the cells with a light miscrope.[1][9][10]
Life cycle
The lifecycle consists of several stadia: gametocyst - oocyst - sporozoite – trophozoite (chepaline) - gamont (gametocyte, sporadin) – gametocyst – oocyst. The gametocyst and oocysts are the cell structures that can survive outside the host-organism and infect other insects. The cycle starts with the oocysts that leave the body with the feces and are left on plant-material. When other locust than eat the plant with oocysts they will burst open (excytation) under the influence of the digestion of the locust. Eight sporozoits will then be released inside the digestive system of the locust.[1][11][12] Orthoptera regurgitate enzymes from the foregut and together with secretions of the salivary glands these enzymes can break down the oocyst cell wall and thus enable the freeing of the sporozoits. The free sporozoits than pass through the peritrophic membrane surrounding the midgut. Once in the ectoperitrophic space they can attach themselves to the epithelial cells of the caeca and midgut.[13][14][15]

Once attached to a epithelial cell, it grows vegetatively and becomes a trophozoite (also called cephalin or chepalont). After 48 hours, a cell with two structures can be seen: the epimerite, attaching to the host cell and the second part (back) of the cell. After a while, a septum is formed, creating a clear protomerite and deuteromerite.[1] After about eight days the Trophozoite will release itself from the host cell.[1][16][17] After release from the epithelial cell, a trophozoit associates with a second one and forms a gamont by forming a circle and fusing together (syzygy).[1][18][19][20][19] Once no distinction between the two is visible, zygotes are formed. The zygotes are the only diploid lifestage in the lifecycle of G. garnhami. A cyst (oocyst) eventually is formed and the nucleus goes through a meiotic and mitotic division. In the end, eight haploid sporozoits are formed, ready to be released into a new host.[1][21]
Relationship with their host
Their relationship with their hosts has not been deciphered in full detail,[22] but G. garnhami is generally regarded as a commensal organism that does not harm its host. A typical characteristic is that most eugregarines only have one host during their development.[3] This relationship is generally described as host-specific, but this can also be due to the lack of research on this topic.[23] This host-specificity is set to take place at the level of a specific family of hosts in the case of Orthoptera hosts.[23][19] Although some studies mention more hosts per species of gregarine.[24] Lifestage specificity has been shown and this is mainly important for holometabolic insects.[25] Due to a lack of genetic information on gregarines it is however difficult to study the host-specificity. The identification of gregarines is often based on external characteristics, but they are very similar making identification difficult.[19][26][27] In the past (and as of 2020) identification of gregarines was often based on the host, but this seems not to be an advisable method.[28]
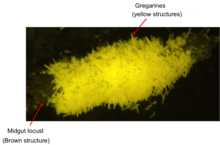
G. garnhami is mainly found in the caeca and midgut of the desert locust where it attaches itself to the epithelial cells in order to feed.[1] The uptake of food happens through the epimerite, attached to the host cell, after a while also the developed protomerite and deutomerite can take up nutrients.[29] The uptake of nutrients happens through osmosis and pinocytosis (formation of a Cytostome). The nutrients are then stored as paraglycogen or fat.[9][30][31][32] The hundreds of epicytic fold give eugregarines a substantial enlargement of the cell-surface in order to take up more nutrients.[33] More details need to be studied in regard of the nutrient uptake by (eu)gregarines, however, it is clear that they do not pierce the host cell to take up the cytoplasm. An apical complex is absent in the trophozoit stage in most eugregarines while it is this complex that is used by archigregarines for the uptake of nutrients by piercing the host cell. [32][29][31][13] G. garnhami stays present as a extracellular organism in the host, not penetrating the cellmenbrane of the host.[2] The effect of G. garnhami on the host is thus minimal. Up to 10 000 gregarines can be present in one host without an effect of the shedding or food uptake of the host.[34]
References
- Canning, Elizabeth U. (1956). "A New Eugregarine of Locusts, Gregarina garnhami n.sp., parasitic in Schistocerca gregaria Forsk". The Journal of Protozoology. 3 (2): 50–62. doi:10.1111/j.1550-7408.1956.tb02433.x. ISSN 1550-7408.
- Valigurová, Andrea; Koudela, Břetislav (August 2008). "Morphological analysis of the cellular interactions between the eugregarine Gregarina garnhami (Apicomplexa) and the epithelium of its host, the desert locust Schistocerca gregaria". European Journal of Protistology. 44 (3): 197–207. doi:10.1016/j.ejop.2007.11.006. PMID 18304787.
- Lange, C.E.; Lord, J.C. (2012). Insect Pathology - 2nd Edition (2nd ed.). Elsevier/Academic Press. pp. 367–387. ISBN 9780123849847.
- Vivier, E; Petitprez, A (November 1969). "The outer membrane complex and its development at the time of the formation of daughter cells in Toxoplasma gondii". The Journal of Cell Biology. 43 (2): 329–42. doi:10.1083/jcb.43.2.329. PMC 2107857. PMID 5344151.
- Walker, M. H.; Mackenzie, C.; Bainbridge, S. P.; Orme, C. (1979). "A Study of the Structure and Gliding Movement of Gregarina garnhami". The Journal of Protozoology. 26 (4): 566–574. doi:10.1111/j.1550-7408.1979.tb04197.x. ISSN 1550-7408.
- Schrevel, J.; Caigneaux, E.; Gros, D.; Philippe, M. (1 May 1983). "The three cortical membranes of the gregarines. I. Ultrastructural organization of Gregarina blaberae". Journal of Cell Science. 61 (1): 151–174. ISSN 0021-9533. PMID 6411745.
- Morrissette, NS; Sibley, LD (March 2002). "Cytoskeleton of apicomplexan parasites". Microbiology and Molecular Biology Reviews. 66 (1): 21–38, table of contents. doi:10.1128/mmbr.66.1.21-38.2002. PMC 120781. PMID 11875126.
- Apicomplexa tolweb.org, Tree of life we project, Jan Šlapeta and Victoria Morin-Adeline
- Vivier, E. (1968). "L'Organisation Ultrastructurale Corticale de la Gregarine Lecudina pellucida; ses Rapports avec l'Alimentation et la Locomotion". The Journal of Protozoology. 15 (2): 230–246. doi:10.1111/j.1550-7408.1968.tb02115.x. ISSN 1550-7408.
- Schrével, Joseph (1 January 1994). "Protists: An exceptional source of cell models". Biology of the Cell. 80 (2): 241–256. doi:10.1111/j.1768-322X.1994.tb00935.x. ISSN 0248-4900.
- Janardanan, K.P.; Ramachandran, P. (1983). "On a new septate gregarine, Stenoductus polydesmi sp. n. (Sporozoa: Cephalina) from the Millipede, Polydesmus sp. with observations on host-specificity of gregarines infecting the millipedes in Kerala, India". Acta Protozoologica. 22 (1): 87–94.
- Thomas, W. P. (1 January 1965). "A Note on the Life Cycle of Eugregarines Inhabiting the Alimentary Canal of Epiphyas Postvittana (walker) (lepidoptera: Tortricidae)". New Zealand Entomologist. 3 (4): 42–46. doi:10.1080/00779962.1965.9722848. ISSN 0077-9962.
- Valigurová, Andrea (2008). Comparative morphology of developmental stages of gregarines and cryptosporidia with an emphasis on host-parasite interactions (Thesis). Masaryk University, Faculty of Science.
- Chapman, R.F. (1998). The insects : structure and function (4th ed.). Cambridge University Press. p. 788. ISBN 9780511818202.
- CLOPTON, RICHARD E.; LUCAROTTI, CHRISTOPHER J. (September 1997). "Leidyana canadensis N. Sp. (Apicomplexa: Eugregarinida) from Larval Eastern Hemlock Looper, Lambdina fiscellaria fiscellaria (Lepidoptera: Geometridae)". The Journal of Eukaryotic Microbiology. 44 (5): 383–387. doi:10.1111/j.1550-7408.1997.tb05712.x.
- Corbel, J.C. (1964). "Infestations experimentales de Locusta migratoria L. (insect, orthoptère) par Gregarina garnhami Canning (sporozoaire Gregarinoporphe): relation entre le cycle de l'hôte et celui du parasite". Comptes rendus des séances de l'Académie des Sciences. 259: 207–210.
- Valigurová, A.; Michalková, V.; Koudela, B. (2009). "Eugregarine trophozoite detachment from the host epithelium via epimeriet retraction: Fiction or fact?". International Journal for Parasitology. 39 (11): 1235–1242. doi:10.1016/j.ijpara.2009.04.009. PMID 19460380.
- Smyth, D.; Wakelin, J.D. (1994). Introduction to animal parasitology (3rd ed.). Cambridge University Press. pp. 88–108. ISBN 9780521428118.
- Clopton, Richard E. (2009). "Phylogenetic Relationships, Evolution, and Systematic Revision of the Septate Gregarines (Apicomplexa: Eugregarinorida: Septatorina)". Comparative Parasitology. 76 (2): 167–190. doi:10.1654/4388.1. ISSN 1525-2647. S2CID 12944487.
- Toso, MA; Omoto, CK (June 2007). "Ultrastructure of the Gregarina niphandrodes nucleus through stages from unassociated trophozoites to gamonts in syzygy and the syzygy junction". The Journal of Parasitology. 93 (3): 479–84. doi:10.1645/GE-1028R1.1. PMID 17626337. S2CID 8444394.
- Harry, Owen G. (6 April 2009). "Studies on infection and reinfection by eugregarines". Parasitology. 63 (2): 213–223. doi:10.1017/S0031182000079531.
- Chapman, R.F.; Joern, A. (1990). Biology of grasshoppers. Wiley. p. 563. ISBN 978-0-471-60901-8.
- Clopton, RE; Gold, RE (May 1996). "Host Specificity of Gregarina blattarum von Siebold, 1839 (Apicomplexa: Eugregarinida) among Five Species of Domiciliary Cockroaches". Journal of Invertebrate Pathology. 67 (3): 219–23. doi:10.1006/jipa.1996.0036. PMID 8812602.
- Corbel, J.C. (1973). "Observations complémentaires sur la repartition géographique et al spécificité parasitaire de Gregarina acridiorum Léger, 1893 (eugregarina, gregarinidae)". Acrida. 2: 69–75.
- Clopton, RE; Janovy J, Jr; Percival, TJ (April 1992). "Host stadium specificity in the gregarine assemblage parasitizing Tenebrio molitor". The Journal of Parasitology. 78 (2): 334–7. doi:10.2307/3283484. JSTOR 3283484. PMID 1556647.
- Clopton, R.E. (2000). An illustrated guide to the protozoa : organisms traditionally referred to as protozoa, or newly discovered groups (2nd ed.). Society of Protozoologists. pp. 205–288. ISBN 1891276239.
- Lange, CE; Wittenstein, E (January 2002). "The life cycle of Gregarina ronderosi n. sp. (Apicomplexa: Gregarinidae) in the Argentine grasshopper Dichroplus elongatus (Orthoptera: Acrididae)". Journal of Invertebrate Pathology. 79 (1): 27–36. doi:10.1016/S0022-2011(02)00008-3. PMID 12054784.
- Cielocha, Joanna J.; Cook, Tamara J.; Clopton, Richard E. (January 2011). "Host Utilization and Distribution of Nubenocephalid Gregarines (Eugregarinorida: Actinocephalidae) Parasitizing Argia spp. (Odonata: Zygoptera) in the Central United States". Comparative Parasitology. 78 (1): 152–160. doi:10.1654/4459.1. S2CID 85731261.
- Kamm, Minnie Elizabeth Watson (NaN). Studies on gregarines, including descriptions of twenty-one new species and a synopsis of the eugregarine records from the Myriapoda, Coleoptera and Orthoptera of the world. University of Illinois. Check date values in:
|date=(help) - Walsh, R. D.; Callaway, C. S. (1969). "The Fine Structure of the Gregarine Lankesteria culicis Parasitic in the Yellow Fever Mosquito Aedes aegypti". The Journal of Protozoology. 16 (3): 536–545. doi:10.1111/j.1550-7408.1969.tb02313.x. ISSN 1550-7408. PMID 4981008.
- MacMillan, W. G. (NaN). "Gregarine attachment organelles — structure and permeability of an interspecific cell junction". Parasitology. 66 (1): 207–214. doi:10.1017/S0031182000044553. ISSN 1469-8161. Check date values in:
|date=(help) - Leander, Brian S. (1 July 2006). "Ultrastructure of the archigregarine Selenidium vivax (Apicomplexa) – A dynamic parasite of sipunculid worms (host: Phascolosoma agassizii)". Marine Biology Research. 2 (3): 178–190. doi:10.1080/17451000600724395. ISSN 1745-1000.
- Leander, Brian S. (2007). "Molecular phylogeny and ultrastructure of Selenidium serpulae (Apicomplexa, Archigregarinia) from the calcareous tubeworm Serpula vermicularis (Annelida, Polychaeta, Sabellida)". Zoologica Scripta. 36 (2): 213–227. doi:10.1111/j.1463-6409.2007.00272.x. ISSN 1463-6409.
- Harry, Owen G. (March 1970). "Gregarines: their Effect on the Growth of the Desert Locust ( Schistocerca gregaria )". Nature. 225 (5236): 964–966. Bibcode:1970Natur.225..964H. doi:10.1038/225964a0. ISSN 1476-4687. PMID 16056839. S2CID 4189137.
External links
| Wikisource has the text of the 1911 Encyclopædia Britannica article Gregarines. |