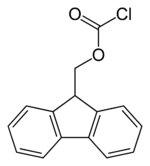
Fluorenylmethyloxycarbonyl chloride
Fluorenylmethyloxycarbonyl chloride (Fmoc-Cl) is a chloroformate ester. It is used to introduce the fluorenylmethyloxycarbonyl protecting group as the Fmoc carbamate.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Chloroformic acid 9H-fluoren-9-ylmethyl ester | |
| Other names
9-Fluorenylmethyl chloroformate; 9-Fluorenylmethoxycarbonyl chloride; Fmoc-chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.044.816 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H11ClO2 | |
| Molar mass | 258.70 g·mol−1 |
| Melting point | 62 to 64 °C (144 to 147 °F; 335 to 337 K) |
| Hazards | |
EU classification (DSD) (outdated) |
Corrosive (C) |
| R-phrases (outdated) | R34 |
| S-phrases (outdated) | S26 S36/37/39 S45 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
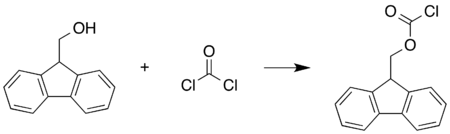
Preparation
This compound may be prepared by reacting 9-fluorenylmethanol with phosgene:[2]
gollark: In some languages.
gollark: I think it's a separate character.
gollark: Is æ a *ligature*?
gollark: The second-most-technologically-advanced on CodersNet!
gollark: We're a real nation on many Minecraft servers!
References
- Fmoc chloride at Sigma-Aldrich
- Carpino, Louis A.; Han, Grace Y. (1972). "9-Fluorenylmethoxycarbonyl amino-protecting group". The Journal of Organic Chemistry. 37 (22): 3404. doi:10.1021/jo00795a005.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.