Ethylene (data page)
This page provides supplementary chemical data on ethylene.
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction | ? |
| Dielectric constant | ? ε0 at ? °C |
| Bond strength | ? |
| Bond length | C-C 133pm C-H 108 pm |
| Bond angle | 121.7 |
| Magnetic susceptibility | ? |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 104 K (−169 °C), 120 Pa |
| Critical point | 282.5 K (9.4 °C), 50.6 bar |
| Std enthalpy change of fusion, ΔfusH |
+3.35 kJ/mol |
| Std entropy change of fusion, ΔfusS |
+32.2 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH |
+13.5 kJ/mol |
| Std entropy change of vaporization, ΔvapS |
? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
117.8 J/(mol K) |
| Heat capacity, cp | 67.4 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
+52.47 kJ/mol |
| Standard molar entropy, S |
219.32 J/(mol K) |
| Enthalpy of combustion, ΔcH |
–1387.4 kJ/mol |
| Heat capacity, cp | 42.9 J/(mol K) |
| van der Waals' constants[1] | a = 453.02 L2 kPa/mol2 b = 0.05714 liter per mole |
Vapor pressure of liquid
| P in mmHg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15200 | 30400 | ||
| T in °C | –168.3 | –153.2 | –141.3 | –131.8 | –113.9 | –103.7 | –90.8 | –71.1 | –32 | –29.1 | –1.5 | — | |
Table data obtained from CRC Handbook of Chemistry and Physics, 44th ed.
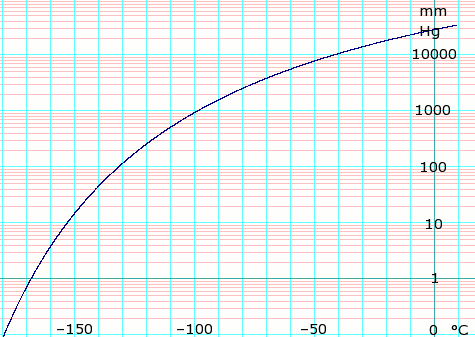
log10 of ethylene vapor pressure. Uses formula: , obtained from Lange's Handbook of Chemistry, 10th ed.
Spectral data
| UV-Vis | |
|---|---|
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| IR | |
| Major absorption bands | 974 cm−1 |
| NMR | |
| Proton NMR | |
| Carbon-13 NMR | |
| Other NMR data | |
| MS | |
| Masses of main fragments |
|
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source such as SIRI, and follow its directions.
gollark: Market systems typically provide more choice about work and whatnot than centrally planned ones.
gollark: https://i.redd.it/ktdfmqa6w4t51.png
gollark: https://i.redd.it/lm5x20x6zft51.jpg
gollark: meme.
gollark: https://i.redd.it/ymlbbzgwsxt51.jpg
References
- Lange's Handbook of Chemistry 10th ed, pp 1522-1524
Except where noted otherwise, data relate to standard ambient temperature and pressure.
Disclaimer applies.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.