Ethyl acetate (data page)
This page provides supplementary chemical data on ethyl acetate.
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommended that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source and follow its directions.
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction, nD | 1.3720 |
| Abbe number | ? |
| Dielectric constant, εr | 6.02 ε0 at 25 °C |
| Bond strength | ? |
| Bond length | ? |
| Bond angle | ? |
| Magnetic susceptibility | ? |
| Surface tension | 23.9 dyn/cm at 20 °C |
| Viscosity[1] | 0.5285 mPa·s at 0 °C 0.4546 mPa·s at 20 °C 0.3668 mPa·s at 40 °C |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 189.3 K (−83.9 °C), ? Pa |
| Critical point | 530 K (260 °C), 3900 kPa |
| Std enthalpy change of fusion, ΔfusH |
10.48 kJ/mol (189.3 K) |
| Std entropy change of fusion, ΔfusS |
55.27 J/(mol·K) (189.3 K) |
| Std enthalpy change of vaporization, ΔvapH |
31.94 kJ/mol |
| Std entropy change of vaporization, ΔvapS |
100.21 J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
−480 kJ/mol |
| Standard molar entropy, S |
259.4 J/(mol K) |
| Heat capacity, cp | 170 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
−445 kJ/mol |
| Standard molar entropy, S |
363 J/(mol K) |
| Heat capacity, cp | 125.8 J/(mol K) (360 K) |
| van der Waals' constants[2] | a = 2072.0 L2 kPa/mol2 b = 0.1412 liter per mole |
Vapor pressure of liquid
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15200 | 30400 | 45600 | |
| T in °C | –43.4 | –13.5 | 9.1 | 27.0 | 59.0 | 77.1 | 100.6 | 136.6 | 169.7 | 209.5 | — | — | |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
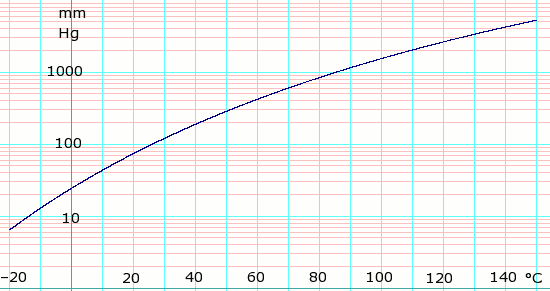
log10 of Ethyl Acetate vapor pressure. Uses formula: obtained from Lange's Handbook of Chemistry, 10th ed.
Distillation data
|
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Spectral data
| UV-Vis | |
|---|---|
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| IR | |
| Major absorption bands | 2983, 1743, 1374, 1243, 1048 cm−1 |
| NMR | |
| Proton NMR | δ CDCl3 4.12 (2H, q), 2.04 (3H, s), 1.26 (3H, t). |
| Carbon-13 NMR | δ CDCl3 171.1, 60.4, 21.0, 14.3. |
| Other NMR data | JH-H (in ethyl), 7.1 Hz. |
| MS | |
| Masses of main fragments |
88.0 (5.1%), 70.0 (9.9%), 61.0 (14.9%), 45.0 (14.7%), 43.0 (100.0%), 29.0 (13.7%) |
gollark: It is unreasonable to go "hmm, yes, these people are pedophiles/supporting them and should be obliterated" because of discussing vaguely adjacent stuff.
gollark: DO NOT us-versus-themize things || 🐝.
gollark: Are NONE safe?
gollark: Do you not have alts within esolangs?
gollark: You could *theoretically* have some complicated system where children do "responsibility tests" or something, but I don't think it would work very well.
References
- Lange's Handbook of Chemistry, 10th ed. pp 1669-1674
- Lange's Handbook of Chemistry 10th ed, pp 1522-1524
- "Binary Vapor-Liquid Equilibrium Data" (Queriable database). Chemical Engineering Research Information Center. Retrieved 6 May 2007.
- Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD), http://webbook.nist.gov
- SDBS spectral database
Except where noted otherwise, data relate to standard ambient temperature and pressure.
Disclaimer applies.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.