Ethane (data page)
This page provides supplementary chemical data on ethane.
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source such as SIRI, and follow its directions.
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction, nD | ? |
| Abbe number | ? |
| Dielectric constant, εr | ? ε0 at ? °C |
| Bond strength | ? |
| Bond length | ? |
| Bond angle | ? |
| Magnetic susceptibility | ? |
| Surface tension | 21.16 dyn/cm at −119.9 °C |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 91 K (−182 °C), 1.1 Pa |
| Critical point | 305.3 K (32.2 °C), 4.9 MPa |
| Std enthalpy change of fusion, ΔfusH |
9.76 kJ/mol at −182 °C |
| Std entropy change of fusion, ΔfusS |
6.46 J/(mol·K) at −182 °C |
| Std enthalpy change of vaporization, ΔvapH crystal I → liquid |
14.703 kJ/mol at −89.0 °C |
| Std entropy change of vaporization, ΔvapS crystal I → liquid |
79.87 J/(mol·K) at −89.0 °C |
| Std enthalpy change of state transition, ΔtrsH crystal II → crystal I |
2.282 kJ/mol at −183.3 °C |
| Std entropy change of state transition, ΔtrsS crystal II → crystal I |
25.48 kJ/mol at −183.3 °C |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
126.7 J/(mol K) |
| Heat capacity, cp | 68.5 J/(mol K) at −179 °C |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
−83.8 kJ/mol |
| Standard molar entropy, S |
229.6 J/(mol K) |
| Enthalpy of combustion, ΔcH |
−1560.7 kJ/mol |
| Heat capacity, cp | 52.49 J/(mol K) at 25 °C |
| van der Waals' constants[1] | a = 556.2 L2 kPa/mol2 b = 0.06380 L/mol |
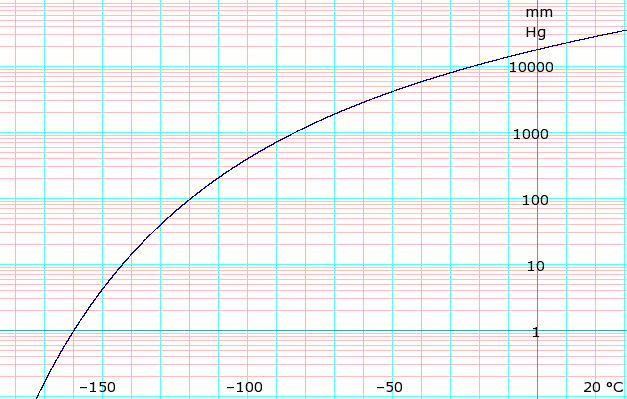
Vapor pressure of liquid
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15200 | 30400 | 45600 | |
| T in °C | −159.5 | −142.9 | −129.8 | −119.3 | −99.6 | −88.6 | −75.0 | −52.8 | −32.0 | −6.4 | 23.6 | — | |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
Melting point data
Mean value for acceptable data: −183.01 °C (90.14 K).
Sources used, from ONS Open Melting Point Collection:[3]
Values considered "outliers", not included in averaging:
Spectral data
| UV-Vis | |
|---|---|
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| IR | |
| Major absorption bands | ? cm−1 |
| NMR | |
| Proton NMR | |
| Carbon-13 NMR | |
| Other NMR data | |
| MS | |
| Masses of main fragments |
|
gollark: Hey, maybe I should rewrite my eternally unfinished project in ReasonML or something. It'll never be finished either way.
gollark: I like the whitespacelessness.
gollark: Why is that different to regular calls?
gollark: Oh, right, you can remap keywords? No, then.
gollark: It looks like you actually *could* handle this with just a JS interpreter with extra primitives, apart from the "states" which are basically closures anyway.
References
- Lange's Handbook of Chemistry 10th ed, pp 1522-1524
- "Pure Component Properties" (Queriable database). Chemical Engineering Research Information Center. Retrieved 8 May 2007.
- "ONS Open Melting Point Collection". 2011. doi:10.1038/npre.2011.6229.1.
- Burnett, Lowell J.; Muller, Burton H. (1970). "Melting points of ethane and three of its deuterated modifications". J. Chem. Eng. Data. 15 (1): 154–157. doi:10.1021/je60044a013.
- "CHERIC". Chemical Engineering Research Information Center. Archived from the original (Queriable database) on 2 August 2002. Retrieved 25 August 2011.
- Givens, F. L.; McCormick, W. D. (1977). "Proton magnetic resonance line shape measurements in solid ethane". J. Chem. Phys. 67: 1150–6. doi:10.1063/1.434967.
- "PHYSPROP" (Queriable database). SRC, Inc. (Syracuse Research Corporation). Retrieved 25 August 2011.
- Witt, R. K.; Kemp, J. D. (1937). "The Heat Capacity of Ethane from 15°K. to the Boiling Point. The Heat of Fusion and the Heat of Vaporization". 59 (2): 273–276. doi:10.1021/ja01281a013. Cite journal requires
|journal=(help) - "Safety data for ethane" (Queriable database). Oxford University. Retrieved 25 August 2011.
- Streng, A. G. (1971). "Miscibility and compatibility of some liquefied and solidified gases at low temperatures". J. Chem. Eng. Data. 16 (3): 357–359. doi:10.1021/je60050a024.
Except where noted otherwise, data relate to standard ambient temperature and pressure.
Disclaimer applies.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.