EPN (insecticide)
EPN is an insecticide of the phosphonothioate class. It is used against pests such as European corn borer, rice stem borer, bollworm, tobacco budworm, and boll weevil.[4]
 | |
| Names | |
|---|---|
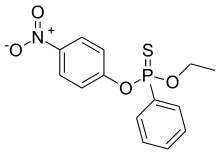
| IUPAC name
O-Ethyl O-(4-nitrophenyl) phenylphosphonothioate | |
| Other names
Ethyl p-nitrophenyl thionobenzenephosphonate, Ethyl p-nitrophenyl benzenethionophosphonate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.016.615 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C14H14NO4PS | |
| Molar mass | 323.30 g·mol−1 |
| Appearance | Light yellow crystalline powder[1] |
| Density | 1.3 g/cm3[1] |
| Melting point | 36 °C (97 °F; 309 K)[1] |
| Boiling point | 215°C at 0.667kPa |
| Insoluble[1] | |
| Hazards | |
| Flash point | noncombustible [2] |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
20 mg/kg (oral, dog) 8 mg/kg (oral, rat) 36 mg/kg (oral, rat) 7 mg/kg (oral, rat) 12.2 mg/kg (oral, mouse)[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 0.5 mg/m3 [skin][2] |
REL (Recommended) |
TWA 0.5 mg/m3 [skin][2] |
IDLH (Immediate danger) |
5 mg/m3[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Available forms
EPN is available in two forms, EPN( O-ethyl O-p-nitrophenyl phenylphosphonothionate) and EPNO (O-ethyl O-p-nitrophenyl phenylphosphonate). EPNO differs from EPN by having an oxygen atom in place of the sulfur atom.
Both these compounds have a (+)- and a (−)-isomer. There is no apparent differences in the rate of hydrolysis of these isomers, but the (+)-isomer of both EPN and EPNO is more toxic to house flies. While the (+)- and (−)-isomers, and the racemic mixture of both isomers of EPN, are equally toxic to mice. The (+)-isomer of EPNO is more toxic to mice than the (−)-EPNO isomer.
Mechanism of action
EPN, via its oxygen analog EPNO generated by metabolism, causes delayed neurotoxicity. It is an acetylcholinesterase (AChE) inhibitor. AChE is an enzyme that hydrolyzes acetylcholine, an excitatory neurotransmitter. When acetylcholine is released into the synaptic cleft, the postsynaptic action is not terminated by reuptake. Rather, the acetylcholine is broken down by AChE into acetate and choline which is then taken up by the presynaptic terminal where the choline together with acetyl CoA is resynthesized into acetylcholine. AChE is therefore present in high concentrations in the synaptic cleft.
EPN can enter the nervous system readily due to it being lipophilic in nature. Here it inhibits AChE by binding to a serine residue located at the active site of AChE. The subsequent lack of acetylcholine hydrolysis causes accumulation of acetylcholine at cholinergic synapses. This in turn causes cholinergic receptors to become overstimulated.
Metabolism
EPN itself is not directly toxic; the phosphorus-sulfur group is biotransformed into a phosphorus-oxygen group. The newly obtained oxygen analog is the compound that inhibits. Furthermore, EPN has been observed to yield different metabolic products in animals, including as p-aminophenyl ethyl benzenethiophosphonate, O-ethyl phenylphosphonic acid, phenylphosphonic acid, O-ethyl phenylphosphonothioic acid, p-nitrophenol, and phenol. The p-nitrophenol can be further metabolized in the liver. The remaining amine is still a weak inhibitor.
Applications
EPN is an insectide and an acaricide effective against orchard pests, including apple flea weevil, plum curculio, and coddling moth and for some soil insects. It is also good to use against the following pests: rice stem borer, boll weevils, oriental fruit moth, fruit moths, codling moths, cotton bollworms, peachtree borers, pear psylla, aphids, scale, budmoths, leafrollers, mites, European cornborrers, aphids, thrips, armyworms, leaf miners, mexican beetles and many others.
Toxicity
In humans, EPN causes various symptoms including sweating, tearing, weakness, headache, dizziness, nausea, vomiting, tightness in chest, seizures, loss of consciousness, diarrhea, and abdominal cramps. The toxicity of EPN has been determined by performing animal experiments on various species, including different types of rodents, different types of birds and even cats and dogs15. EPN has been administered to the animal test subjects via different ways of exposure including oral-, skin-, eye-, intraperitoneal- and subcutaneous administration>. Exposure to humans can happen through inhalation of the aerosol, ingestion and absorption through skin. There is no reliable information available on LD50 values in humans. However, the recommended limits of skin exposure to EPN, stated by the American Conference of Governmental Industrial Hygienists (ACGIH) in 2008, were a time weighted average of 0.5 mg/m3 and a short term exposure level of 2 mg/m3. Additionally when human volunteers were fed 6 mg of EPN per day for 47 days, no effect was found. When the daily dose was raised to 9 mg for 57 days, a reversible inhibition of blood cholinesterase was found. In table 1 some LD50 values for various non-human species are listed.
Table 1: LD50 values of EPN for different routes of exposure administered to various organisms
All data was derived from16
| Route of exposure | Organism | LD50 value |
| Oral | Chicken | 5 mg/kg bodyweight |
| Dog | 20 mg/kg bodyweight | |
| Duck | 3 mg/kg bodyweight | |
| Mouse | 12.2 mg/kg bodyweight | |
| Pigeon | 4.21 mg/kg bodyweight | |
| Quail | 5 mg/kg bodyweight | |
| Rat | 7 mg/kg bodyweight | |
| Wild bird | 2.37 mg/kg bodyweight | |
| Inhalation | Rat | 106 mg/m3/1H |
| Skin | Cat | 45 mg/kg |
| Duck | 400 mg/kg | |
| Mouse | 348 mg/kg | |
| Rabbit | 30 mg/kg | |
| Rat | 25 mg/kg |
Acute toxicity
When rats inhaled a lethal dose of EPN they showed muscle twitches (fasciculations) originating from the peripheral nervous system. Furthermore convulsions of the lungs and thorax were observed along with shortness of breath. However when a lethal dose of EPN was administered via the skin to rats, the rats showed excitement additional to the aforementioned effects. When EPN was administered to cats via the skin, similar behavior was observed, although the animals showed signs of paralysis without anesthesia instead of fasciculations. Overall it can be stated that an overdose of EPN, results in convulsions and tremor and eventually causes dead. The period till death after administration of a lethal dose of EPN is, however, not listed.
Occupational toxicity
Different countries have set different limits of acceptable occupational toxicity of EPN. These values range from 0.1-0.5 mg/m3 via skin exposure. Chronic effects were not observed when hen were fed 18 ppm EPN for 21 months. When fed 54 ppm for 21 months signs of delayed neurotoxicity were observed.
Treatment
There is no specific antidote listed for EPN. Symptoms of acute poisoning by EPN develop during exposure or in the following twelve hours. These symptoms range from dizziness and headaches, to tremor, muscle twitching and if exposed to a high dose even to respiratory deficiency. Since EPN is an organophosphorus compound that inhibits acetylcholinesterase (AChE), it is advised to administer atropine sulfite intravenously or intramusculary until atropinization is achieved. This atropinization can take up to 12 hours and must be repeated in order to have the desired effect.
References
- "EPN International Chemical Safety Card". National Institute for Occupational Safety and Health. Archived from the original on 2017-12-05. Retrieved 2017-09-09.
- NIOSH Pocket Guide to Chemical Hazards. "#0255". National Institute for Occupational Safety and Health (NIOSH).
- "EPN". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). 4 December 2014. Retrieved 19 March 2015.
- "EPN Chemical Profile". Pesticide Management Education Program, Cornell University.
External links
- EPN, toxipedia.org
- NIOSH Pocket Guide to Chemical Hazards, cdc.gov