Dihydroxybenzenes
Dihydroxybenzenes are organic chemical compounds in which two hydroxyl groups are substituted onto a benzene ring. These aromatic compounds are classed as phenols. There are three isomer: 1,2-dihydroxybenzene (the ortho isomer) is commonly known as catechol, 1,3-dihydroxybenzene (the meta isomer) is commonly known as resorcinol, and 1,4-dihydroxybenzene (the para isomer) is commonly known as hydroquinone.[1]
ortho
isomermeta
isomerpara
isomerCatechol
pyrocatechol
1,2-dihydroxybenzene
o-dihydroxybenzene
1,2-benzenediol
o-benzenediolResorcinol
1,3-dihydroxybenzene
m-dihydroxybenzene
resorcin
1,3-benzenediol
m-benzenediolHydroquinone
1,4-dihydroxybenzene
p-dihydroxybenzene
1,4-benzenediol
p-benzenediol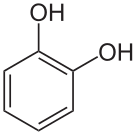
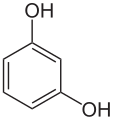
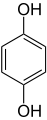
All three of these compounds are colorless to white granular solids at room temperature and pressure, but upon exposure to oxygen they may darken. All three isomers have the chemical formula C6H6O2.
Similar to other phenols, the hydroxyl groups on the aromatic ring of a benzenediol are weakly acidic. Each benzenediol can lose an H+ from one of the hydroxyls to form a type of phenolate ion.
The Dakin oxidation is an organic redox reaction in which an ortho- or para-hydroxylated phenyl aldehyde or ketone reacts with hydrogen peroxide in base to form a benzenediol and a carboxylate. Overall, the carbonyl group is oxidized, and the hydrogen peroxide is reduced.
See also
References
- Helmut Fiege, Heinz-Werner Voges, Toshikazu Hamamoto, Sumio Umemura, Tadao Iwata, Hisaya Miki, Yasuhiro Fujita, Hans-Josef Buysch, Dorothea Garbe, Wilfried Paulus (2002). "Phenol Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_313. ISBN 978-3527306732.CS1 maint: uses authors parameter (link)