Cyclam
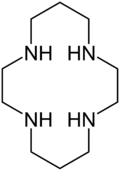
Cyclam (1,4,8,11-tetraazacyclotetradecane) is an organic compound with the formula (NHCH2CH2NHCH2CH2CH2)2. It is a white solid that is soluble in water. The compound is notable as a macrocyclic ligand, which binds strongly to many transition metal cations.[1] The compound was first prepared by the reaction of 1,3-dibromopropane and ethylenediamine.[2]
Cl2.png)
 | |
 | |
| Names | |
|---|---|
| IUPAC name
1,4,8,11-Tetraazacyclotetradecane | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.491 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H24N4 | |
| Molar mass | 200.330 g·mol−1 |
| Melting point | 185 to 188 °C (365 to 370 °F; 458 to 461 K) |
| 5 g/100 mL (20 °C) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The compound features four secondary amines. Its complexes therefore can exist as several diastereomers, depending on the relative orientation of the N–H centres. Its complexes feature alternating five- and six-membered chelate rings. The closely related ligand cyclen ((CH2CH2NH)4) forms only five-membered C2N2M chelate rings and tends not to form square-planar complexes.
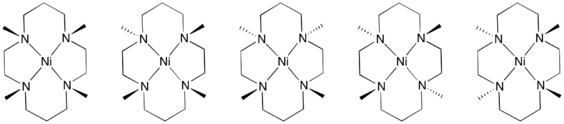
N-Alkyl derivatives
Metal-cyclam complexes are prone to oxidative degradation, which is initiated by deprotonation of the secondary amine. This flaw led to the development of cyclam derivatives wherein the NH centres are replaced by tertiary amines. For example, the tetramethyl derivatives are readily prepared by methylation using formaldehyde and formic acid.[1] These oxidatively robust derivatives of cyclam have enabled a number of metal–O2 complexes.[4]
See also
References
- Barefield, E. Kent (2010). "Coordination chemistry of N-tetraalkylated cyclam ligands—A status report". Coord. Chem. Rev. 254 (15–16): 1607–1627. doi:10.1016/j.ccr.2010.03.007.
- Van Alphen, J. (1937). "On Aliphatic Polyamines IV". Rec. Trav. Chim. Pays-Bas. 56 (4): 343–350. doi:10.1002/recl.19370560405.
- Ito, T.; Kato, M.; Ito, H. (1984). "The Structures of trans-Dichloro- and trans-Bis(isothiocyanato)nickel(II) Complexes with 1,4,8,11-Tetraazacyclotetradecane, 1,4,8,12-Tetraazacyclopentadecane, and 1,5,9,13-Tetraazacyclohexadecane. The Negative Correlation between the Axial and In-plane Coordination Bond Lengths in Tetragonal Ni(II) Complexes of the trans-NiX2N4 Type". Bull. Chem. Soc. Jpn. 57 (9): 2641. doi:10.1246/bcsj.57.2641.
- Cho, J.; Sarangi, R.; Nam, W. (2012). "Mononuclear Metal–O2 Complexes Bearing Macrocyclic N-Tetramethylated Cyclam Ligands". Acc. Chem. Res. 45 (8): 1321–1330. doi:10.1021/ar3000019. PMC 3627547. PMID 22612523.