Butyrate
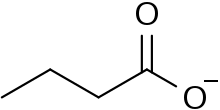
Butyrate or butanoate is the traditional name for the conjugate base of butyric acid (also known as butanoic acid). The formula of the butyrate ion is C
4H
7O−
2. The name is used as part of the name of esters and salts of butyric acid
 | |
| Names | |
|---|---|
| IUPAC name
butanoate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C 4H 7O− 2 | |
| Molar mass | 87.098 g mol−1 |
| Appearance | oily, colorless liquid |
| Density | 0.959 at 20 °C/4 °C |
| Melting point | -7.9 °C[1] |
| Boiling point | 163.5 °C[1] |
| Hazards | |
| Flash point | 72 °C[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
In nature
As a short chain fatty acid, butyric acid is fully ionized near neutral pH. In nature, the cation associated with butyrate is unknown or unimportant to the metabolizing enzymes.
Butyrates are important as food for cells lining the mammalian colon (colonocytes). Without butyrates for energy, colon cells undergo autophagy (self digestion) and die.[2] Short-chain fatty acids (SCFAs), which include butyrate, are produced by beneficial colonic bacteria (probiotics) that feed on, or ferment prebiotics, which are plant products that contain adequate amounts of dietary fiber. These SCFAs benefit the colonocytes (cells of the colon) by increasing energy production and may protect against colon cancer by inhibiting cell proliferation.[3]
Butyrate is a major metabolite in colonic lumen arising from bacterial fermentation of dietary fiber and has been shown to be a critical mediator of the colonic inflammatory response. In fact, butyrate is responsible for about 70% of energy from the colonocytes, being a critical SCFA in the colon homeostasis.[4] Butyrate possesses both preventive and therapeutic potential to counteract inflammation-mediated ulcerative colitis (UC) and colorectal cancer. The reason why butyrate is an energy source for normal colonocytes and induces apoptosis in colon cancer cells, is due to the Warburg effect in cancer cells, which leads to butyrate not being properly metabolized. This phenomenon leads to the accumulation of butyrate in the nucleus, acting as a histone deacetylase (HDAC) inhibitor.[5] One mechanism underlying butyrate function in suppression of colonic inflammation is inhibition of the IFN-γ/STAT1 signaling pathways at least partially through acting as a HDAC inhibitor. It has been shown that butyrate inhibits activity of HDAC1 that is bound to the Fas gene promoter in T cells, resulting in hyperacetylation of the Fas promoter and upregulation of Fas receptor on the T cell surface. It is thus suggested that butyrate enhances apoptosis of T cells in the colonic tissue and thereby eliminates the source of inflammation (IFN-γ production).[6] Butyrate inhibits angiogenesis by inactivating Sp1 transcription factor activity and downregulating vascular endothelial growth factor gene expression.[7]
As a Human Metabolite and Immune Regulator
Butyrate is extremely essential to host immune homeostasis. Although the role and importance of butyrate in the gut is not fully understood, many researchers argue that a depletion of butyrate-producing bacteria in patients with several vasculitic conditions is essential to the pathogenesis of these disorders.
A depletion of butyrate in the gut is typically caused by an absence or depletion of butyrate-producing-bacteria (BPB). This depletion in BPB leads to microbial dysbiosis. This dysbiosis is characterized by an overall low biodiversity and a depletion of key butyrate-producing members. Butyrate is an essential microbial metabolite with a vital role as a modulator of proper immune function in the host. It's been shown that children lacking in BPB are more susceptible to allergic disease [8] and Type 1 Diabetes [9]. Butyrate—the conjugate base of butyric acid—exerts a key role for the maintenance of immune homeostasis both local (in the gut) and systemic (via circulating butyrate). Butyrate has shown to promote the differentiation of regulatory T cells. In particular, circulating butyrate prompts the generation of extrathymic regulatory T cells. The low-levels of butyrate in human subjects could favor reduced regulatory T cell-mediated control, thus promoting a powerful immuno-pathological T-cell response.[10] On the other hand, gut butyrate has been reported to inhibit local pro-inflammatory cytokines. The absence or depletion of these BPB in the gut could therefore be a possible aide in the overly-active inflammatory response. Butyrate in the gut also protects the integrity of the intestinal epithelial barrier. Decreased butyrate levels therefore lead to a damaged or dysfunctional intestinal epithelial barrier.[11]
In a 2013 research study conducted by Furusawa et al, microbe-derived butyrate was found to be essential in inducing the differentiation of colonic regulatory T cells in mice. This is of great importance and possibly relevant to the pathogenesis and vasculitis associated with many inflammatory diseases because regulatory T cells have a central role in the suppression of inflammatory and allergic responses [12]. In several research studies, it has been demonstrated that butyrate induced the differentiation of regulatory T cells in vitro and in vivo.[13]
The anti-inflammatory capacity of butyrate has been extensively analyzed and supported by many studies. It has been found that microorganism-produced butyrate expedites the production of regulatory T cells. An increase in regulatory T cell numbers was observed upon a butyrate provision.[14] More recently, it has been shown that butyrate plays an essential and direct role in modulating gene expression of cytotoxic T-cells.[15] Although specific mechanism by which butyrate aids in the differentiation of T-cells is unclear, it has been determined that butyrate promotes the generation of regulatory T cells.
Potential Treatments for Butyrate Restoration
Due to the importance of butyrate as an inflammatory regulator and immune system contributor, butyrate depletions could be a key factor influencing the pathogenesis of many vasculitic conditions. Because of this, it is essential to harvest healthy levels of butyrate in the gut. Butyrate provisions or fecal microbiota transplants (to restore BPB and symbiosis in the gut) could be effective in replenishing butyrate levels. A fecal microbiota transplant, also known as fecal bacteriotherapy, is the process of restoring bacteria commonly found in the digestive tract with an infusion of feces from a donor. While the description of this may disgust some, fecal bacteriotherapy may quickly and efficiently restore symbiosis in imbalanced microbiomes. In this treatment option, a healthy individual would donate their stool to be transplanted into an individual with dysbiosis. Another less-invasive treatment option that may be effective in decreasing inflammation in individuals with vasculitic conditions is the administration of butyrate. This could be as easy as buying oral butyrate supplements online. Another way to administer butyrate into the gut—which may be more effective though also more invasive—is by the administration of butyrate enemas. Butyrate—both oral supplements and enemas—has been shown to be very effective in terminating symptoms of inflammation with minimal-to-no side-effects. In a study where patients with ulcerative colitis were treated with butyrate enemas, inflammation decreased significantly, and bleeding ceased completely after butyrate provision.[16] Butter is a natural source of Butyrate.
Synthetic butyrates
A variety of butyrate esters are known. Examples include:
- Cellulose acetate butyrate, an aircraft dope
- Methyl butyrate
- Ethyl butyrate
- Butyl butyrate
- Pentyl butyrate
Salts of butyrates are also well known. One example is sodium butyrate, a colorless, water-soluble salt. It is a histone deacetylase inhibitor (HDAC) used in psychiatry.[17]
See also
References
- "Butyrate".
- Donohoe, Dallas R.; Garge, Nikhil; Zhang, Xinxin; et al. (2011). "The Microbiome and Butyrate Regulate Energy Metabolism and Autophagy in the Mammalian Colon". Cell Metabolism. 13 (5): 517–26. doi:10.1016/j.cmet.2011.02.018. PMC 3099420. PMID 21531334.
- Lupton, Joanne R. (February 2004). "Microbial Degradation Products Influence Colon Cancer Risk: the Butyrate Controversy". J. Nutr. 134 (2): 479–482.
- Zeng, Huawei; Lazarova, DL; Bordonaro, M (2014). "Mechanisms linking dietary fiber, gut microbiota and colon cancer prevention". World Journal of Gastrointestinal Oncology. 6 (2): 41–51. doi:10.4251/wjgo.v6.i2.41. PMC 3926973. PMID 24567795.
- Encarnação, J. C.; Abrantes, A. M.; Pires, A. S.; et al. (30 July 2015). "Revisit dietary fiber on colorectal cancer: butyrate and its role on prevention and treatment". Cancer and Metastasis Reviews. 34 (3): 465–478. doi:10.1007/s10555-015-9578-9. PMID 26224132.
- Zimmerman, Mary A.; Singh, Nagendra; Martin, Pamela M.; et al. (15 June 2012). "Butyrate suppresses colonic inflammation through HDAC1-dependent Fas upregulation and Fas-mediated apoptosis of T cells". American Journal of Physiology. Gastrointestinal and Liver Physiology. 302 (12): G1405–G1415. doi:10.1152/ajpgi.00543.2011. PMC 3378095. PMID 22517765.
- Prasanna Kumar, S.; Thippeswamy, G.; Sheela, M.L.; et al. (October 2008). "Butyrate-induced phosphatase regulates VEGF and angiogenesis via Sp1". Archives of Biochemistry and Biophysics. 478 (1): 85–95. doi:10.1016/j.abb.2008.07.004. PMID 18655767.
- Cait, Alissa; Cardenas, Erick (December 2019). "Reduced genetic potential for butyrate fermentation in the gut microbiome of infants who develop allergic sensitization". Journal of Allergy and Clinical Immunology. 144 (6): 1638-1647. E3. doi:10.1016/j.jaci.2019.06.029.
- Vatanen, T.; Franzosa, E.A.; Schwager, R.; et al. (2018). "The human gut microbiome in early-onset type 1 diabetes from the TEDDY study". Nature. 562: 589–594. doi:10.1038/s41586-018-0620-2.
- Consolandi, Clarissa; Turroni, Silvia; Emmi, Giacomo; et al. (April 2015). "Behçet's syndrome patients exhibit specific microbiome signature". Autoimmunity Reviews. 14 (4): 269–276. doi:10.1016/j.autrev.2014.11.009. PMID 25435420.
- Ye, Zi; Zhang, Ni; Wu, Chunyan; et al. (4 August 2018). "A metagenomic study of the gut microbiome in Behcet's disease". Microbiome. 6 (1): 135. doi:10.1186/s40168-018-0520-6. PMC 6091101. PMID 30077182.
- Cait, Alissa; Hughes, Michael R Hughes (May 2018). "Microbiome-driven allergic lung inflammation is ameliorated by short chain fatty acids". Mucosal Immunology. 11 (3): 785–796. doi:10.1038/mi.2017.75.
- Furusawa, Yukihiro; Obata, Yuuki; Fukuda, Shinji; et al. (13 November 2013). "Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells". Nature. 504 (7480): 446–450. Bibcode:2013Natur.504..446F. doi:10.1038/nature12721. PMID 24226770.
- Arpaia, Nicholas; Campbell, Clarissa; Fan, Xiying; et al. (13 November 2013). "Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation". Nature. 504 (7480): 451–455. Bibcode:2013Natur.504..451A. doi:10.1038/nature12726. PMC 3869884. PMID 24226773.
- Luu, Maik; Weigand, Katharina; Wedi, Fatana; et al. (26 September 2018). "Regulation of the effector function of CD8+ T cells by gut microbiota-derived metabolite butyrate". Scientific Reports. 8 (1): 14430. doi:10.1038/s41598-018-32860-x. PMC 6158259. PMID 30258117.
- Scheppach, W.; Sommer, H.; Kirchner, T.; et al. (1992). "Effect of butyrate enemas on the colonic mucosa in distal ulcerative colitis". Gastroenterology. 103: 51–56. doi:10.1016/0016-5085(92)91094-K.
- Peedicayil, Jacob (2017). Epigenetics in Psychiatry. Elsevier. ISBN 978-0-12-810108-7. OCLC 968290293.