Aziridine
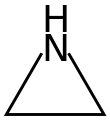
Aziridine is an organic compound consisting of the three-membered heterocycle (CH2)2NH.[5][6] It is a colorless, toxic, volatile liquid that is of significant practical interest.[7] Its derivatives, also referred to as aziridines, are of broader interest in medicinal chemistry.
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Aziridine | |||
| Systematic IUPAC name
Azacyclopropane | |||
| Other names
Azirane Ethylene imine Aminoethylene Dimethyleneimine Dimethylenimine Ethylimine | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 102380 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.268 | ||
| EC Number |
| ||
| 616 | |||
| KEGG | |||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1185 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C2H5N | |||
| Molar mass | 43.069 g·mol−1 | ||
| Appearance | Colorless oily liquid[1] | ||
| Odor | ammonia-like[2] | ||
| Density | 0.8321 g/mL 20 °C[3] | ||
| Melting point | −77.9 °C (−108.2 °F; 195.2 K) | ||
| Boiling point | 56 °C (133 °F; 329 K) | ||
| miscible | |||
| Vapor pressure | 160 mmHg (20° C)[2] | ||
| Hazards | |||
| Main hazards | highly flammable and toxic | ||
| GHS pictograms |      | ||
| GHS Signal word | Danger | ||
GHS hazard statements |
H225, H300, H310, H314, H318, H330, H340, H350, H411 | ||
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P262, P264, P270, P271, P273, P280, P281, P284, P301+310, P301+330+331, P302+350, P303+361+353, P304+340, P305+351+338, P308+313, P310 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −11 °C (12 °F; 262 K) | ||
| 322 °C (612 °F; 595 K) | |||
| Explosive limits | 3.6–46% | ||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration) |
250 ppm (rat, 1 hr) 250 ppm (guinea pig, 1 hr) 62 ppm (rat, 4 hr) 223 ppm (mouse, 2 hr) 56 ppm (rat, 2 hr) 2236 ppm (mouse, 10 min)[4] | ||
LCLo (lowest published) |
25 ppm (guinea pig, 8 hr) 56 ppm (rabbit, 2 hr)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
OSHA-Regulated Carcinogen[2] | ||
REL (Recommended) |
Ca[2] | ||
IDLH (Immediate danger) |
Ca [100 ppm][2] | ||
| Related compounds | |||
Related heterocycles |
Borirane Ethylene oxide Thiirane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Structure
The bond angles in aziridine are approximately 60°, considerably less than the normal hydrocarbon bond angle of 109.5°, which results in angle strain as in the comparable cyclopropane and ethylene oxide molecules. A banana bond model explains bonding in such compounds. Aziridine is less basic than acyclic aliphatic amines, with a pKa of 7.9 for the conjugate acid, due to increased s character of the nitrogen free electron pair. Angle strain in aziridine also increases the barrier to nitrogen inversion. This barrier height permits the isolation of separate invertomers, for example the cis and trans invertomers of N-chloro-2-methylaziridine.
Synthesis and uses
Aziridine is produced industrially from aminoethanol via two related routes. The Nippon Shokubai process requires an oxide catalyst and high temperatures to effect the dehydration. In the Wenker synthesis, the aminoethanol is converted to the sulfate ester, which undergoes base-induced sulfate elimination. Older methods entailed amination of 1,2-dichloroethane and cyclization of 2-chloroethylamine.[7]
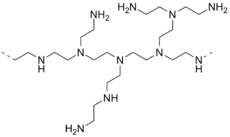
Aziridine forms a wide variety of polymeric derivatives, known as polyethylenimines. These and related species are useful crosslinking agents and precursors for coatings.[7]
Safety
Aziridine is highly toxic with an LD50 of 14 mg (oral, rats). It is a skin irritant. As an alkylating agent, it also a mutagen.[7] It is subject to attack and ring-opening by endogenous nucleophiles such as nitrogenous bases in DNA base pairs, resulting in potential mutagenicity.[8][9][10]
See also
- Binary ethylenimine, a dimeric form of aziridine
References
- "Aziridine" (PDF). Re-evaluation of Some Organic Chemicals, Hydrazine and Hydrogen Peroxide. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. 71. 1999.
- NIOSH Pocket Guide to Chemical Hazards. "#0274". National Institute for Occupational Safety and Health (NIOSH).
- Weast, Robert C.; et al. (1978). CRC Handbook of Chemistry and Physics (59th ed.). West Palm Beach, FL: CRC Press. ISBN 0-8493-0549-7.
- "Ethyleneimine". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- Gilchrist, T.L. Heterocyclic chemistry. ISBN 978-0-582-01421-3.
- Epoxides and aziridines – A mini review Albert Padwa, S. Shaun Murphree Arkivoc (JC-1522R) pp. 6–33 Online article
- Steuerle, Ulrich; Feuerhake, Robert (2006). "Aziridines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_239.pub2.
- Kanerva L, Keskinen H, Autio P, Estlander T, Tuppurainen M, Jolanki R (May 1995). "Occupational respiratory and skin sensitization caused by polyfunctional aziridine hardener". Clin Exp Allergy. 25 (5): 432–9. doi:10.1111/j.1365-2222.1995.tb01074.x. PMID 7553246.
- Sartorelli P, Pistolesi P, Cioni F, Napoli R, Sisinni AG, Bellussi L, Passali GC, Cherubini Di Simplicio E, Flori L (2003). "Skin and respiratory allergic disease caused by polyfunctional aziridine". Med Lav. 94 (3): 285–95. PMID 12918320.
- Mapp CE (2001). "Agents, old and new, causing occupational asthma". Occup. Environ. Med. 58 (5): 354–60. doi:10.1136/oem.58.5.354. PMC 1740131. PMID 11303086.
| Wikimedia Commons has media related to Aziridine. |