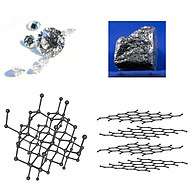
Allotropy
Allotropy or allotropism (from Ancient Greek ἄλλος (allos), meaning 'other', and τρόπος (tropos), meaning 'manner, form') is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element;[1] the atoms of the element are bonded together in a different manner. For example, the allotropes of carbon include diamond (the carbon atoms are bonded together in a tetrahedral lattice arrangement), graphite (the carbon atoms are bonded together in sheets of a hexagonal lattice), graphene (single sheets of graphite), and fullerenes (the carbon atoms are bonded together in spherical, tubular, or ellipsoidal formations). The term allotropy is used for elements only, not for compounds. The more general term, used for any crystalline material, is polymorphism. Allotropy refers only to different forms of an element within the same phase (i.e.: solid, liquid or gas states); the differences between these states would not alone constitute examples of allotropy.
For some elements, allotropes have different molecular formulae despite difference in phase; for example, two allotropes of oxygen (dioxygen, O2, and ozone, O3) can both exist in the solid, liquid and gaseous states. Other elements do not maintain distinct allotropes in different phases; for example, phosphorus has numerous solid allotropes, which all revert to the same P4 form when melted to the liquid state.
History
The concept of allotropy was originally proposed in 1841 by the Swedish scientist Baron Jöns Jakob Berzelius (1779–1848).[2] The term is derived from Greek άλλοτροπἱα (allotropia), meaning 'variability, changeableness'.[3] After the acceptance of Avogadro's hypothesis in 1860, it was understood that elements could exist as polyatomic molecules, and two allotropes of oxygen were recognized as O2 and O3.[2] In the early 20th century, it was recognized that other cases such as carbon were due to differences in crystal structure.
By 1912, Ostwald noted that the allotropy of elements is just a special case of the phenomenon of polymorphism known for compounds, and proposed that the terms allotrope and allotropy be abandoned and replaced by polymorph and polymorphism.[2] Although many other chemists have repeated this advice, IUPAC and most chemistry texts still favour the usage of allotrope and allotropy for elements only.[4]
Differences in properties of an element's allotropes
Allotropes are different structural forms of the same element and can exhibit quite different physical properties and chemical behaviours. The change between allotropic forms is triggered by the same forces that affect other structures, i.e., pressure, light, and temperature. Therefore, the stability of the particular allotropes depends on particular conditions. For instance, iron changes from a body-centered cubic structure (ferrite) to a face-centered cubic structure (austenite) above 906 °C, and tin undergoes a modification known as tin pest from a metallic form to a semiconductor form below 13.2 °C (55.8 °F). As an example of allotropes having different chemical behaviour, ozone (O3) is a much stronger oxidizing agent than dioxygen (O2).
List of allotropes
Typically, elements capable of variable coordination number and/or oxidation states tend to exhibit greater numbers of allotropic forms. Another contributing factor is the ability of an element to catenate.
Examples of allotropes include:
Non-metals
| Element | Allotropes |
|---|---|
| Carbon |
|
| Phosphorus |
|
| Oxygen |
|
| Sulfur |
|
| Selenium |
|
Metalloids
| Element | Allotropes |
|---|---|
| Boron |
|
| Silicon |
|
| Arsenic |
|
| Germanium |
|
| Antimony |
|
| Tellurium |
|
Metals
Among the metallic elements that occur in nature in significant quantities (56 up to U, without Tc and Pm), almost half (27) are allotropic at ambient pressure: Li, Be, Na, Ca, Ti, Mn, Fe, Co, Sr, Y, Zr, Sn, La, Ce, Pr, Nd, Sm, Gd, Tb, Dy, Yb, Hf, Tl, Th, Pa and U. Some phase transitions between allotropic forms of technologically relevant metals are those of Ti at 882 °C, Fe at 912 °C and 1394 °C, Co at 422 °C, Zr at 863 °C, Sn at 13 °C and U at 668 °C and 776 °C.
| Element | Phase name(s) | Space group | Pearson symbol | Structure type | Description |
|---|---|---|---|---|---|
| Lithium | R3m | hR9 | α-Samarium structure | Forms below 70 K.[6] | |
| Im3m | cI2 | Body-centered cubic | Stable at room temperature and pressure. | ||
| cF4 | Face centered cubic | Forms above 7GPa | |||
| hR1 | An intermediate phase formed ~40GPa. | ||||
| cI16 | Forms above 40GPa. | ||||
| Beryllium | P63/mmc | hP2 | Hexagonal close packed | Stable at room temperature and pressure. | |
| Im3m | cI2 | Body centered cubic | Forms above 1255°C. | ||
| Sodium | R3m | hR9 | α-Samarium structure | Forms below 20 K. | |
| Im3m | cI2 | Body centered cubic | Stable at room temperature and pressure. | ||
| Fm3m | cF4 | Face centered cubic | Forms at room temperature above 65 GPa.[7] | ||
| I43d | cI16 | Forms at room temperature, 108GPa.[8] | |||
| Pnma | oP8 | Forms at room temperature, 119GPa.[9] | |||
| Magnesium | P63/mmc | hP2 | hexagonal close packed | Stable at room temperature and pressure. | |
| Im3m | cI2 | Body centered cubic | Forms above 50 GPa.[10] | ||
| Tin | α-tin, gray tin, tin pest | Fd3m | cF8 | Diamond cubic | Stable below 13.2 °C. |
| β-tin, white tin | I41/amd | tI4 | β-Tin structure | Stable at room temperature and pressure. | |
| γ-tin, rhombic tin | I4/mmm | Body-centered tetragonal | |||
| σ-Sn | Body-centered cubic | Forms at very high pressure.[11] | |||
| Stanene | |||||
| Iron | α-Fe, ferrite | Im3m | cI2 | Body-centered cubic | Stable at room temperature and pressure. Ferromagnetic at T<770 °C, paramagnetic from T=770–912 °C. |
| γ-iron, austenite | Fm3m | cF4 | Face-centered cubic | Stable from 912–1,394 °C. | |
| δ-iron | Im3m | cI2 | Body-centered cubic | Stable from 1,394 – 1,538 °C, same structure as α-Fe. | |
| ε-iron, Hexaferrum | P63/mmc | hP2 | Hexagonal close-packed | Stable at high pressures. | |
| Cobalt | α-Cobalt | simple cubic | Forms above 417 °C. | ||
| β-Cobalt | hexagonal close packed | Forms below 417 °C. | |||
| Polonium | α-Polonium | simple cubic | |||
| β-Polonium | rhombohedral |
Lanthanides and actinides
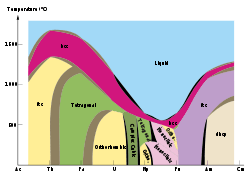
- Cerium, samarium, dysprosium and ytterbium have three allotropes.
- Praseodymium, neodymium, gadolinium and terbium have two allotropes.
- Plutonium has six distinct solid allotropes under "normal" pressures. Their densities vary within a ratio of some 4:3, which vastly complicates all kinds of work with the metal (particularly casting, machining, and storage). A seventh plutonium allotrope exists at very high pressures. The transuranium metals Np, Am, and Cm are also allotropic.
- Promethium, americium, berkelium and californium have three allotropes each.[12]
Nanoallotropes
In 2017, the concept of nanoallotropy was proposed by Prof. Rafal Klajn of the Organic Chemistry Department of the Weizmann Institute of Science.[13] Nanoallotropes, or allotropes of nanomaterials, are nanoporous materials that have the same chemical composition (e.g., Au), but differ in their architecture at the nanoscale (that is, on a scale 10 to 100 times the dimensions of individual atoms).[14] Such nanoallotropes may help create ultra-small electronic devices and find other industrial applications.[14] The different nanoscale architectures translate into different properties, as was demonstrated for surface-enhanced Raman scattering performed on several different nanoallotropes of gold.[13] A two-step method for generating nanoallotropes was also created.[14]
See also
- Isomer
- Polymorphism (materials science)
- Superdense carbon allotrope
Notes
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "Allotrope". doi:10.1351/goldbook.A00243
- Jensen, W. B. (2006), "The Origin of the Term Allotrope", J. Chem. Educ., 83 (6): 838–39, Bibcode:2006JChEd..83..838J, doi:10.1021/ed083p838.
- "allotropy", A New English Dictionary on Historical Principles, 1, Oxford University Press, 1888, p. 238.
- Jensen 2006, citing Addison, W. E. The Allotropy of the Elements (Elsevier 1964) that many have repeated this advice.
- Raj, G. Advanced Inorganic Chemistry Vol-1. Krishna Prakashan. p. 1327. ISBN 9788187224037. Retrieved January 6, 2017.
- Overhauser, A. W. (1984-07-02). "Crystal Structure of Lithium at 4.2 K". Physical Review Letters. American Physical Society (APS). 53 (1): 64–65. doi:10.1103/physrevlett.53.64. ISSN 0031-9007.
- Hanfland, M.; Loa, I.; Syassen, K. (2002-05-13). "Sodium under pressure: bcc to fcc structural transition and pressure-volume relation to 100 GPa". Physical Review B. American Physical Society (APS). 65 (18). doi:10.1103/physrevb.65.184109. ISSN 0163-1829.
- McMahon, M. I.; Gregoryanz, E.; Lundegaard, L. F.; Loa, I.; Guillaume, C.; Nelmes, R. J.; Kleppe, A. K.; Amboage, M.; Wilhelm, H.; Jephcoat, A. P. (2007-10-18). "Structure of sodium above 100 GPa by single-crystal x-ray diffraction". Proceedings of the National Academy of Sciences. Proceedings of the National Academy of Sciences. 104 (44): 17297–17299. doi:10.1073/pnas.0709309104. ISSN 0027-8424.
- Gregoryanz, E.; Lundegaard, L. F.; McMahon, M. I.; Guillaume, C.; Nelmes, R. J.; Mezouar, M. (2008-05-23). "Structural Diversity of Sodium". Science. American Association for the Advancement of Science (AAAS). 320 (5879): 1054–1057. doi:10.1126/science.1155715. ISSN 0036-8075.
- Olijnyk, H.; Holzapfel, W. B. (1985-04-01). "High-pressure structural phase transition in Mg". Physical Review B. American Physical Society (APS). 31 (7): 4682–4683. doi:10.1103/physrevb.31.4682. ISSN 0163-1829.
- Molodets, A. M.; Nabatov, S. S. (2000). "Thermodynamic Potentials, Diagram of State, and Phase Transitions of Tin on Shock Compression". High Temperature. 38 (5): 715–721. doi:10.1007/BF02755923.
- Benedict, U.; Haire, R. G.; Peterson, J. R.; Itie, J. P. (1985). "Delocalisation of 5f electrons in curium metal under high pressure". Journal of Physics F: Metal Physics. 15 (2): L29–L35. Bibcode:1985JPhF...15L..29B. doi:10.1088/0305-4608/15/2/002.
- Udayabhaskararao, Thumu; Altantzis, Thomas; Houben, Lothar; Coronado-Puchau, Marc; Langer, Judith; Popovitz-Biro, Ronit; Liz-Marzán, Luis M.; Vuković, Lela; Král, Petr (2017-10-27). "Tunable porous nanoallotropes prepared by post-assembly etching of binary nanoparticle superlattices". Science. 358 (6362): 514–518. Bibcode:2017Sci...358..514U. doi:10.1126/science.aan6046. ISSN 0036-8075. PMID 29074773.
- "Materials That Don't Exist in Nature Might Lead to New Fabrication Techniques". israelbds.org. Archived from the original on 2017-12-09. Retrieved 2017-12-08.
References
- Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica (11th ed.). Cambridge University Press.
External links
| Wikimedia Commons has media related to Allotropy. |
- Nigel Bunce and Jim Hunt. "The Science Corner: Allotropes". Archived from the original on January 31, 2008. Retrieved January 6, 2017.CS1 maint: BOT: original-url status unknown (link)
- Allotropes – Chemistry Encyclopedia