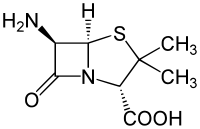
6-APA
6-APA is an abbreviation used for the name of the chemical compound (+)-6-aminopenicillanic acid. In 1958, Beecham scientists found a way to obtain 6-APA from penicillin.[1] Other β-lactam antibiotics could then be synthesized by attaching various side-chains to the nucleus. The other antibiotics are used in place of penicillin G or penicillin V .[2]
 | |
.gif) | |
| Names | |
|---|---|
| IUPAC name
(2S,5R,6R)-6-Amino-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.177 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H12N2O3S | |
| Molar mass | 216.26 g·mol−1 |
| Appearance | colourless |
| Melting point | 198 °C (388 °F; 471 K) |
| 0.4 g/100 mL | |
| log P | 0.600 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The reason why this was achieved so many years after the commercial development of penicillin by Howard Florey and Ernst Chain lies in the fact that penicillin itself is very susceptible to hydrolysis, so direct replacement of the side-chain was not a practical route to other β-lactam antibiotics.
References
- Batchelor, F. R.; Doyle, F. P.; Nayler, J. H. C.; Rolinson, G. N. (1959). "Synthesis of Penicillin: 6-Aminopenicillanic Acid in Penicillin Fermentations". Nature. 183 (4656): 257–258. Bibcode:1959Natur.183..257B. doi:10.1038/183257b0. PMID 13622762.
- F.P. Doyle, J.H.C. Nayler, G.N. Rolinson US Patent 2,941,995, filed July 22, 1958, granted June 21, 1960. Recovery of solid 6-aminopenicillanic acid.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.