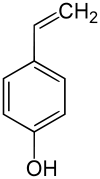
4-Vinylphenol
4-Vinylphenol is a phenolic compound found in wine and beer. It is produced by the spoilage yeast Brettanomyces. When it reaches concentrations greater than the sensory threshold, it can give the wine aromas described as barnyard, medicinal, band-aids, and mousy. In wine, 4-vinylphenol can react with other molecules, such as anthocyanidins, to produce new chemical compounds.[1] In white wines vinylphenols are dominant (4-vinylphenol 70-1 150 μg/l, 4-vinylguaiacol 10-490 μg/l) whereas, in red wines, it is the corresponding ethyl phenols.[2]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
4-Ethenylphenol | |
| Other names
p-Vinylphenol p-Hydroxystyrene 4-VP | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.018.276 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H8O | |
| Molar mass | 120.15 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Biochemistry
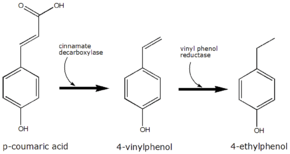
4-Ethylphenol is produced from the precursor p-coumaric acid. Brettanomyces converts this to 4-vinylphenol via the enzyme cinnamate decarboxylase.[3] 4-Vinylphenol is further reduced to 4-ethylphenol by the enzyme vinyl phenol reductase. Coumaric acid is sometimes added to microbiological media, enabling the positive identification of Brettanomyces by smell.
See also
- 4-Ethylguaiacol
- Yeast in winemaking
- Wine fault
- Wine chemistry
- Poly-4-vinylphenol (polyvinylphenol or PVP) is a plastic structurally similar to polystyrene.
References
- Structure of new anthocyanin-derived wine pigments. Hélène Fulcrand, Paulo-Jorge Cameira dos Santos, Pascale Sarni-Manchado, Véronique Cheynier and Jean Favre-Bonvin, J. Chem. Soc., Perkin Trans. 1, 1996, pages 735-739, doi:10.1039/P19960000735
- Flüchtige phenolische Verbindungen in Wein = Volatil phenolic compounds of wine. Rapp A. and Versini G., Deutsche Lebensmittel-Rundschau, 1996, vol. 92, no2, pages 42-48, INIST:2990913 (German)
- Brettanomyces Monitoring by Analysis of 4-ethylphenol and 4-ethylguaiacol Archived 2008-02-19 at the Wayback Machine at etslabs.com