Homoisoflavonoid
Homoisoflavonoids (3-benzylidenechroman-4-ones) are a type of phenolic compounds[1] occurring naturally in plants.
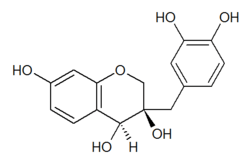
Chemically, they have the general structure of a 16-carbon skeleton, which consists of two phenyl rings (A and B) and heterocyclic ring (C).
Synthesis
Homoisoflavones can be synthetized from 2'-hydroxydihydrochalcones.[2]
Homoisoflavanones can be synthetized[3] from 3,5-methoxy phenols via chroman-4-one in three steps[4] or from phloroglucinol.[5]
- Conversion
Homoisoflavanes can be obtained from the conversion of homoisoflavonoids.[6]
Natural occurrences
The homoisoflavonoids portulacanones A, B, C and D can be found in Portulaca oleracea (common purslane, Caryophyllales, Portulacaceae).[7]
The 3,4-dihydroxyhomoisoflavans sappanol, episappanol, 3'-deoxysappanol, 3'-O-methylsappanol and 3'-O-methylepisappanol can be found in Caesalpinia sappan.[8]
The homoisoflavones scillavones A and B can be isolated from the bulbs of Scilla scilloides (Barnardia japonica).[9]
Homoisoflavanones
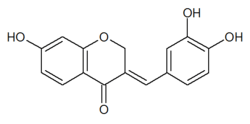
Homoisoflavanones (3-Benzyl-4-chromanones[10]) can be found in various plants,[11] notably in Hyacinthaceae (Scilloideae).[12]
Sappanone A can be found in Caesalpinia sappan.[13]
C-Methylated homoisoflavanones (3-(4'-methoxy-benzyl)-5,7-dihydroxy-6-methyl-8-methoxy-chroman-4-one, 3-(4'-methoxy-benzyl)-5,7-dihydroxy-6,8-dimethyl-chroman-4-one, 3-(4'-hydroxy-benzyl)-5,7-dihydroxy-6,8-dimethyl-chroman-4-one, 3-(4'-hydroxy-benzyl)-5,7-dihydroxy-6-methyl-8-methoxy-chroman-4-one and 3-(4'-hydroxy-benzyl)-5,7-dihydroxy-6-methyl-chroman-4-one) can be found in the rhizomes of Polygonum odoratum.[14]
5,7-Dihydroxy-3-(3-hydroxy-4-methoxybenzyl)-chroman-4-one, a homoisoflavanone extracted from Cremastra appendiculata (Orchidaceae), has anti-angiogenic activities and inhibits UVB-induced skin inflammation through reduced cyclooxygenase-2 expression and NF-?B nuclear localization.[15]
In Asparagaceae
3-(4'-Methoxybenzyl)-7,8-methylenedioxy-chroman-4-one, a homoisoflavanone with antimycobacterial activity, can be isolated from Chlorophytum inornatum (Asparagaceae, Agavoideae).[6]
5,7-Dihydroxy-3-(4-methoxybenzyl)-chroman-4-one, 7-hydroxy-3-(4-hydroxybenzyl)-chroman-4-one and 4’-demethyl-3,9-dihydro-punctatin can be isolated from Agave tequilana (Asparagaceae, Agavoideae).[16]
- in Scilloideae (Hyacinthaceae)
7-O-α-Rhamnopyranosyl-(1→6)-β-glucopiranosyl-5-hydroxy-3-(4-methoxybenzyl)-chroman-4-one, 7-O-α-rhamnopyranosyl-(1→6)-β-glucopiranosyl-5-hydroxy-3-(4′-hydroxybenzyl)-chroman-4-one, 5,7-dihydroxy-3-(4′-methoxybenzyl)-chroman-4-one (3,9-dihidroeucomin), 5,7-dihydroxy-6-methoxy-3-(4′-methoxybenzyl)-chroman-4-one, 5,7-dihydroxy 3-(4′-hydroxybenzyl)-chroman-4-one (4,4′-demethyl-3,9-dihydropuctatin), 5,7-dihydroxy-3-(4′-hydroxybenzyl)-6-methoxy-chroman-4-one (3,9-dihydroeucomnalin) and 7-hydroxy-3-(4′-hydroxybenzyl)-5-methoxy-chroman-4-one can be isolated from the bulbs of Ledebouria floribunda (tribe Hyacintheae).[17] Other compounds can be found in Ledebouria revoluta, a plant widely used as an ethnomedicinal in southern Africa.[10]
The homoisoflavanone glycosides (-)-7-O-methyleucomol 5-O-beta-D-glucopyranoside, (-)-7-O-methyleucomol 5-O-beta-rutinoside and (-)-7-O-methyleucomol 5-O-beta-neohesperidoside can be isolated from the bulbs of Ornithogalum caudatum (tribe Ornithogaloideae).[18]
Scillascillin-type homoisoflavanones (3-hydroxy-type homoisoflavonoids) can be isolated from Drimiopsis maculata (tribe Hyacintheae, Massoniinae).[19]
Eucomin, eucomol,[20] (E)-7-O-methyl-eucomin, (—)-7-O-methyleucomol, (+)-3,9-dihydro-eucomin and 7-O-methyl-3,9-dihydro-eucomin[21] can be isolated from the bulbs of Eucomis bicolor (tribe Hyacintheae, Massoniinae). 4′-o-Methyl-punctatin, autumnalin and 3,9-dihydro-autumnalin can be found in Eucomis autumnalis.[22]
Five homoisoflavanones, 3,5-dihydroxy-7,8-dimethoxy-3-(3',4'-dimethoxybenzyl)-4-chromanone, 3,5-dihydroxy-7-methoxy-3-(3',4'-dimethoxybenzyl)-4-chromanone, 3,5-dihydroxy-7,8-dimethoxy-3-(3'-hydroxy-4'-methoxybenzyl)-4-chromanone, 3,5,6-trihydroxy-7-methoxy-3-(3'-hydroxy-4'-methoxybenzyl)-4-chromanone and 3,5,7-trihydroxy-3-(3'-hydroxy-4'methoxybenzyl)-4-chromanone, can be isolated from the dichloromethane extract of the bulbs of Pseudoprospero firmifolium (tribe Hyacintheae, subtribe Pseudoprospero).[23]
A homoisoflavanone can also be found in Albuca fastigiata (tribe Ornithogaleae).[24]
The same molecule, 5,6-dimethoxy-7-hydroxy-3-(4′-hydroxybenzyl)-4-chromanone, can be found in the bulbs of Resnova humifusa and Eucomis montana (tribe Hyacintheae, subtribe Massoniinae).[25]
Uses
The homoisoflavonoids portulacanones A, B, C and D show in vitro cytotoxic activities towards four human cancer cell lines.[7]
See also
- Flavonoids and isoflavonoids, related chemicals with a 15-carbon skeleton
References
- Roshanak Namdar and Shohreh Nafisi. Study on the interaction of homoisoflavonoids with nucleic acids Comparative study by spectroscopic methods. ISBN 978-3-659-49924-1.
- "New efficient synthesis and bioactivity of homoisoflavonoids". Arkivoc. 2008 (11): 285–294. 2008. doi:10.3998/ark.5550190.0009.b28.
- Jain, Amolak C.; Anita Mehta (née Sharma), (Mrs) (1985). "A new synthesis of homoisoflavanones (3-benzyl-4-chromanones)". Tetrahedron. 41 (24): 5933–5937. doi:10.1016/S0040-4020(01)91433-4.
- Shaikh, Mahidansha; Petzold, Katja; Kruger, Hendrik G.; Du Toit, Karen (2010). "Synthesis and NMR elucidation of homoisoflavanone analogues". Structural Chemistry. 22: 161–166. doi:10.1007/s11224-010-9703-x.
- Rawal, Viresh H.; Cava, Michael P. (1983). "Synthesis of scillascillin, a naturally occurring benzocyclobutene". Tetrahedron Letters. 24 (50): 5581–5584. doi:10.1016/S0040-4039(00)94146-7.
- Zhang, L; Zhang, W. G.; Kang, J; Bao, K; Dai, Y; Yao, X. S. (2008). "Synthesis of (+/-) homoisoflavanone and corresponding homoisoflavane". Journal of Asian Natural Products Research. 10 (9–10): 909–913. doi:10.1080/10286020802217499. PMID 19003606.
- Yan, Jian; Sun, Li-Rong; Zhou, Zhong-Yu; Chen, Yu-Chan; Zhang, Wei-Min; Dai, Hao-Fu; Tan, Jian-Wen (2012). "Homoisoflavonoids from the medicinal plant Portulaca oleracea". Phytochemistry. 80: 37–41. doi:10.1016/j.phytochem.2012.05.014. PMID 22683318.
- Namikoshi, Michio; Nakata, Hiroyuki; Yamada, Hiroyuki; Nagai, Minako; Saitoh, Tamotsu (1987). "Homoisoflavonoids and related compounds. II. Isolation and absolute configurations of 3,4-dihydroxylated homoisoflavans and brazilins from Caesalpinia sappan L". Chemical & Pharmaceutical Bulletin. 35 (7): 2761–2773. doi:10.1248/cpb.35.2761.
- Nishida, Y; Eto, M; Miyashita, H; Ikeda, T; Yamaguchi, K; Yoshimitsu, H; Nohara, T; Ono, M (2008). "A new homostilbene and two new homoisoflavones from the bulbs of Scilla scilloides". Chemical & Pharmaceutical Bulletin. 56 (7): 1022–5. doi:10.1248/cpb.56.1022. PMID 18591825.
- Moodley, N.; Crouch, N.R.; Mulholland, D.A; Slade, D.; Ferreira, D. (2006). "3-Benzyl-4-chromanones (homoisoflavanones) from bulbs of the ethnomedicinal geophyte Ledebouria revoluta (Hyacinthaceae)". South African Journal of Botany. 72 (4): 517–520. doi:10.1016/j.sajb.2006.01.004.
- Du Toit, Karen; Drewes, Siegfried E.; Bodenstein, Johannes (2010). "The chemical structures, plant origins, ethnobotany and biological activities of homoisoflavanones". Natural Product Research. 24 (5): 457–490. doi:10.1080/14786410903335174. PMID 20306368.
- Du Toit, K.; Elgorashi, E.E.; Malan, S.F.; Mulholland, D.A.; Drewes, S.E.; Van Staden, J. (2007). "Antibacterial activity and QSAR of homoisoflavanones isolated from six Hyacinthaceae species". South African Journal of Botany. 73 (2): 236–241. doi:10.1016/j.sajb.2007.01.002.
- Chang, T. S.; Chao, S. Y.; Ding, H. Y. (2012). "Melanogenesis Inhibition by Homoisoflavavone Sappanone a from Caesalpinia sappan". International Journal of Molecular Sciences. 13 (8): 10359–10367. doi:10.3390/ijms130810359. PMC 3431864. PMID 22949866.
- Wang, D; Li, D; Zhu, W; Peng, P (2009). "A new C-methylated homoisoflavanone and triterpenoid from the rhizomes of Polygonatum odoratum". Natural Product Research. 23 (6): 580–9. doi:10.1080/14786410802560633. PMID 19384735.
- Hur, Seulgi; Lee, Yun Sang; Yoo, Hyun; Yang, Jeong-Hee; Kim, Tae-Yoon (2010). "Homoisoflavanone inhibits UVB-induced skin inflammation through reduced cyclooxygenase-2 expression and NF-κB nuclear localization". Journal of Dermatological Science. 59 (3): 163–169. doi:10.1016/j.jdermsci.2010.07.001. PMID 20724116.
- Morales-Serna, José Antonio; Jiménez, Armando; Estrada-Reyes, Rosa; Marquez, Carmen; Cárdenas, Jorge; Salmón, Manuel (2010). "Homoisoflavanones from Agave tequilana Weber". Molecules. 15 (5): 3295–3301. doi:10.3390/molecules15053295. PMC 6263332. PMID 20657479.
- Calvo, María Isabel (2009). "Homoisoflavanones from Ledebouria floribunda". Fitoterapia. 80 (2): 96–101. doi:10.1016/j.fitote.2008.10.006. PMID 19027834.
- Tang, Y; Yu, B; Hu, J; Wu, T; Hui, H (2002). "Three new homoisoflavanone glycosides from the bulbs of Ornithogalum caudatum". Journal of Natural Products. 65 (2): 218–20. doi:10.1021/np010466a. PMID 11858761.
- Koorbanally, C; Crouch, N. R.; Mulholland, D. A. (2001). "Scillascillin-type homoisoflavanones from Drimiopsis maculata (Hyacinthaceae)". Biochemical Systematics and Ecology. 29 (5): 539–541. doi:10.1016/s0305-1978(00)00073-9. PMID 11274776.
- Heller, W.; Tamm, Ch. (1981). "Homoisoflavanones and Biogenetically Related Compounds". Fortschritte der Chemie organischer Naturstoffe / Progress in the Chemistry of Organic Natural Products. Fortschritte der Chemie organischer Naturstoffe / Progress in the Chemistry of Organic Natural Products. 40. pp. 105–152. doi:10.1007/978-3-7091-8611-4_3. ISBN 978-3-7091-8613-8.
- Heller, Werner; Andermatt, Paul; Schaad, Werner A.; Tamm, Christoph (1976). "Homoisoflavanone. IV. Neue Inhaltsstoffe der Eucomin-Reihe von Eucomis bicolor". Helvetica Chimica Acta. 59 (6): 2048–2058. doi:10.1002/hlca.19760590618. PMID 1017955.
- Sidwell, W.T.L.; Tamm, Ch. (1970). "The homo-isoflavones II1). Isolation and structure of 4′-o-methyl-punctatin, autumnalin and 3,9-dihydro-autumnalin". Tetrahedron Letters. 11 (7): 475–478. doi:10.1016/0040-4039(70)89003-7.
- Koorbanally, C; Sewjee, S; Mulholland, D. A.; Crouch, N. R.; Dold, A (2007). "Homoisoflavanones from Pseudoprospero firmifolium of the monotypic tribe Pseudoprospereae (Hyacinthaceae: Hyacinthoideae)". Phytochemistry. 68 (22–24): 2753–6. doi:10.1016/j.phytochem.2007.08.005. PMID 17884116.
- Koorbanally, Chantal; Mulholland, Dulcie A.; Crouch, Neil R. (2005). "A novel 3-hydroxy-3-benzyl-4-chromanone-type homoisoflavonoid from Albuca fastigiata (Ornithogaloideae: Hyacinthaceae)". Biochemical Systematics and Ecology. 33 (5): 545–549. doi:10.1016/j.bse.2004.08.009.
- Koorbanally, Neil A.; Crouch, Neil R.; Harilal, Avinash; Pillay, Bavani; Mulholland, Dulcie A. (2006). "Coincident isolation of a novel homoisoflavonoid from Resnova humifusa and Eucomis montana (Hyacinthoideae: Hyacinthaceae)". Biochemical Systematics and Ecology. 34 (2): 114–118. doi:10.1016/j.bse.2005.08.003.