2,4-Dinitroaniline
2,4-Dinitroaniline is a chemical compound with a formula of C6H5N3O4. It is used as an explosive and as a reagent to detect and characterize aldehydes and ketones.
 The ball-and-stick structure of 2,4-dinitroaniline | |
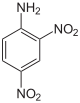 The chemical structure of 2,4-dinitroaniline | |
| Names | |
|---|---|
| IUPAC name
2,4-Dinitroaniline | |
| Other names
1-Amino-2,4-dinitrobenzene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.322 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1596 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H5N3O4 | |
| Molar mass | 183.123 g·mol−1 |
| Appearance | Colorless to yellowish combustible powder |
| Density | 1.61 g/cm |
| Melting point | 187.8 °C (370.0 °F; 460.9 K) |
| Boiling point | Decomposes |
| 0.06 g/L (20 ºC) | |
| Solubility | Soluble in acetone, ethyl acetate, acetonitrile and most alcohols |
| Acidity (pKa) | -4.53 (conjugate acid) ; 18.46 |
| Explosive data | |
| Shock sensitivity | Low |
| Friction sensitivity | Low |
| Detonation velocity | 4,800 m/s [note 1] |
| Hazards | |
| Main hazards | Explosive, Toxic, Health and Environmental hazards |
| GHS pictograms |      |
| GHS Signal word | Danger |
GHS hazard statements |
H300, H310, H330, H373, H411 |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+310, P302+350, P304+340, P310, P314, P320, P321, P322, P330, P361, P363, P391, P403+233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 224 °C (435 °F; 497 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
285 mg/kg (oral, rat) |
| Related compounds | |
Related compounds |
2,4,6-Trinitroaniline |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
2,4-Dinitroaniline can be prepared by reaction of 1-chloro-2,4-dinitrobenzene with ammonia.
It can be also prepared by the electrophilic aromatic substitution of aniline. Direct nitration should not be used due to the reactivity of aniline. (i.e. It can be protonated to anilinium or oxidized easily.) Instead the acetyl protection should be used.
Basicity
Compared to aniline, the basicity of 2,4-dinitroaniline is even weaker. It is due to the electron-withdrawing nature of the nitro groups. This makes the pKa of conjugate acid of 2,4-dinitroaniline being even lower than that of hydronium ions, meaning that it is a strong acid.
The protons in the amino group is also much more acidic than that of aniline.
Uses
2,4-Dinitroaniline is usually used as an explosive. It is also used for the manufacture of azo dyes and disperse dyes, but also can be used as printing ink, toner and preparation of preservatives. and it is an intermediate for disperse dyes, neutral dyes, sulfur dyes, organic pigments.
Safety
2,4-Dinitroaniline is moderately toxic, with a lethal dose of 285 mg/kg. However, the main danger is that it is explosive and flammable with heat or friction.
See also
References
- "2,4-Dinitroaniline CAS#: 97-02-9". www.chemicalbook.com. Retrieved 2019-03-23.
- PubChem. "2,4-Dinitroaniline". pubchem.ncbi.nlm.nih.gov. Retrieved 2019-03-23.
note 1 Calculated from the detonation velocity of 2,4,6-trinitroaniline by multiplying 2/3
